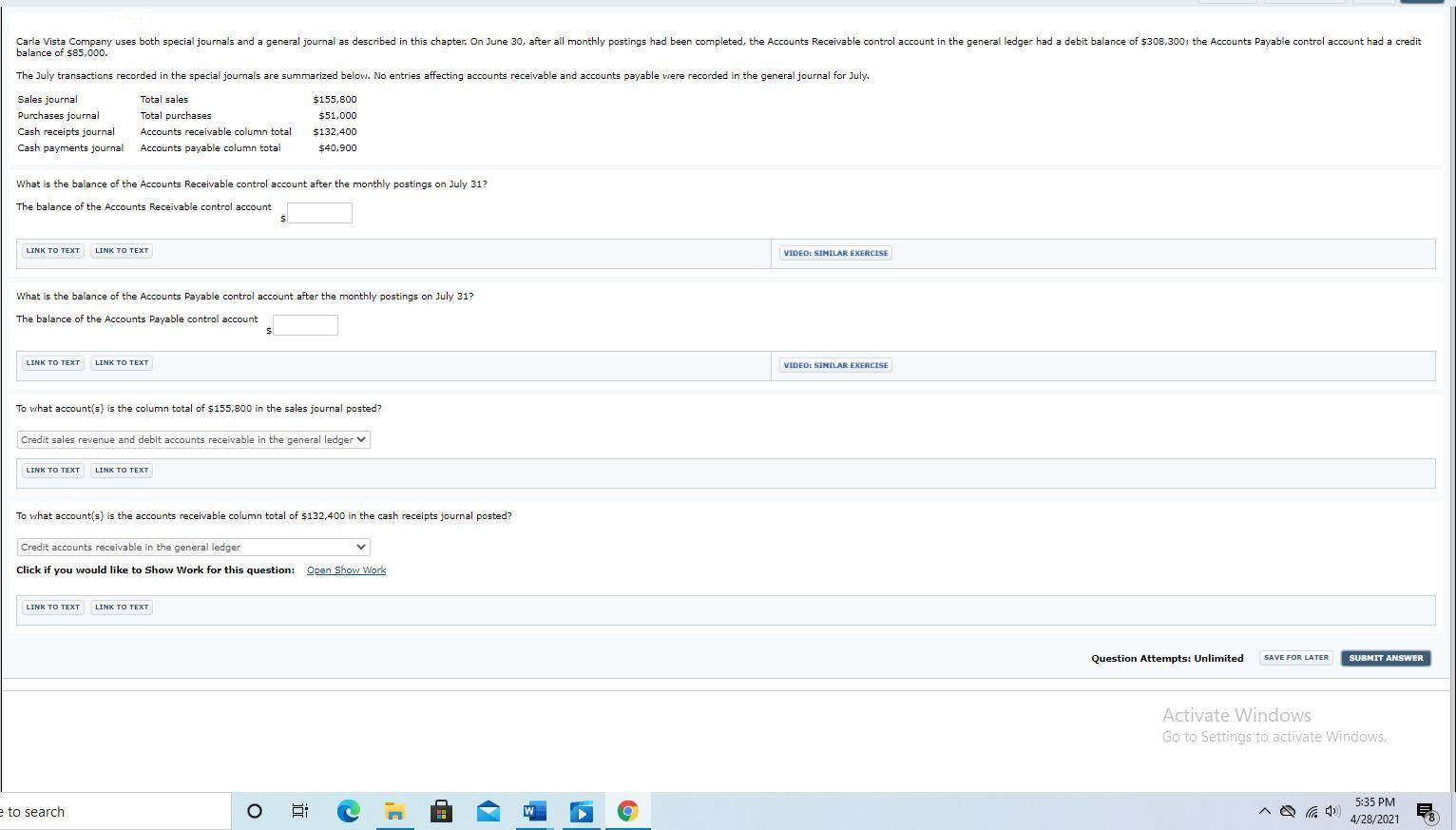
Carla Vista Company uses both special journals and a general journal as described in this chapter....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Carla Vista Company uses both special journals and a general journal as described in this chapter. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in the general ledger had a debit balance of $308,300; the Accounts Payable control account had a credit balance of $85,000. The July transactions recorded in the special journals are summarized below. No entries affecting accounts receivable and accounts payable were recorded in the general journal for July. Sales journal Total sales $155,800 Purchases journal Total purchases $51,000 Cash receipts journal Accounts receivable column total $132,400 Cash payments journal Accounts payable column total $40,900 What is the balance of the Accounts Receivable control account after the monthly postings on July 31? The balance of the Accounts Receivable control account LINK TO TEXT LINK TO TEXT VIDEO: SIMILAR EXERCISE What is the balance of the Accounts Payable control account after the monthly postings on July 31? The balance of the Accounts Payable control account LINK TO TEXT LINK TO TEXT VIDEO: SIMILAR EXERCISE To what account(s) is the column total of $155,800 in the sales journal posted? Credit sales revenue and debit accounts receivable in the general ledger v LINK TO TEXT LINK TO TEXT To what account(s) is the accounts receivable column total of $132,400 in the cash receipts journal posted? Credit accounts receivable in the general ledger Click if you would like to Show Work for this question: Open Show Work LINK TO TEXT LINK TO TEXТ Question Attempts: Unlimited SAVE FOR LATER SUBMIT ANSWER Activate Windows Go to Settings to activate Windows, 5:35 PM e to search Hi 4/28/2021 近 Carla Vista Company uses both special journals and a general journal as described in this chapter. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in the general ledger had a debit balance of $308,300; the Accounts Payable control account had a credit balance of $85,000. The July transactions recorded in the special journals are summarized below. No entries affecting accounts receivable and accounts payable were recorded in the general journal for July. Sales journal Total sales $155,800 Purchases journal Total purchases $51,000 Cash receipts journal Accounts receivable column total $132,400 Cash payments journal Accounts payable column total $40,900 What is the balance of the Accounts Receivable control account after the monthly postings on July 31? The balance of the Accounts Receivable control account LINK TO TEXT LINK TO TEXT VIDEO: SIMILAR EXERCISE What is the balance of the Accounts Payable control account after the monthly postings on July 31? The balance of the Accounts Payable control account LINK TO TEXT LINK TO TEXT VIDEO: SIMILAR EXERCISE To what account(s) is the column total of $155,800 in the sales journal posted? Credit sales revenue and debit accounts receivable in the general ledger v LINK TO TEXT LINK TO TEXT To what account(s) is the accounts receivable column total of $132,400 in the cash receipts journal posted? Credit accounts receivable in the general ledger Click if you would like to Show Work for this question: Open Show Work LINK TO TEXT LINK TO TEXТ Question Attempts: Unlimited SAVE FOR LATER SUBMIT ANSWER Activate Windows Go to Settings to activate Windows, 5:35 PM e to search Hi 4/28/2021 近
Expert Answer:

Related Book For 

Accounting Principles
ISBN: 978-0470533475
9th Edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Posted Date:
Students also viewed these accounting questions
-
Donahue Company uses both special journals and a general journal as described in this chapter. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in...
-
Ruz Company uses both special journals and a general journal as described in this chapter. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in the...
-
Kimani Company uses both special journals and a general journal as described in this appendix. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in...
-
Figure (a) shows a mobile hanging from a ceiling; it consists of two metal pieces (m 1 = 3.5) kg and m 2 = 4.5kg) that are strung together by cords of negligible mass. What is the tension in?? (a)...
-
I. Ertugrul et al. conducted a study to determine the association between insulin growth factor 1 (IGF-1) and bone mineral density (BMD) in men over 65 years of age. The researchers published their...
-
In Section 4.8.1, we considered the use of prefiltering to avoid aliasing. In practice, the antialiasing filter cannot be ideal. However, the nonideal characteristics can be at least partially...
-
Are there any cases you can find where a supplemental pleading was allowed because new facts have been alleged? What were the new facts?
-
Interest Rate Swap On January l, 2013, Marshall Corp. issues $10,000,000 in 4 percent fixed rate debt with interest payments due every six months. Concurrently, Marshall enters into a plain vanilla...
-
You are standing some distance away from two speakers that are arranged one in front of the other and in front of you. They emit identical sound waves. As the speaker farther away from you moves 40...
-
Joe and Jessie are married and have one dependent child, Lizzie. Lizzie is currently in college at State University. Joe works as a design engineer for a manufacturing firm while Jessie runs a craft...
-
The following selected transactions apply to Topeca Supply for November and December, Year 1. November was the first month of operations. Sales tax is collected at the time of sale but is not paid to...
-
In the UCCJEA as outlined in the attached picture, what is the difference between track 3 and track 4, circled in the picture? Why does track 3 require significant connection/substantial evidence and...
-
Set up the following ledger accounts using the account numbers indicated. All accounts will require only one sheet, marked Sheet 1 , except Bank, which will require two ledger sheets. Bank, 1 0 1 ; A...
-
Partial balance sheets and additional Information are listed below for Sowell Company. Sowell Company Partial Balance Sheets as of December 31 Assets Cash Accounts receivable Inventory Liabilities...
-
Two divers are exploring the bottom of a trench in the Pacific Ocean. Dominic is at 171 feet below the surface of the ocean and Liz is at 111 feet below the surface. Represent each quantity by an...
-
Question 5: Working capital What are the components of working capital? What is the cash operating cycle? What techniques might a small business use to manage its stock/inventory levels? What are the...
-
(10 points) Marketing Message: Describe the message you plan to communicate to the target market. This should be a focused message that explains the benefits of the product/event in a way that...
-
If the amplifier indicated by the box input impedance of oo, which of the following statements are true ? has an open loop gain as well as Feedback factor (\beta = 1/ R_1\) The feedback is voltage...
-
Betty Dillman is an accounting major at a midwestern state university located approximately 60 miles from a major city. Many of the students attending the university are from the metropolitan area...
-
The following financial information is available for Cheney Corporation. The weighted average number of shares of common stock outstanding was 80,000 for 2009 and 100,000 for...
-
Matt Reiss owns the Fredonia Barber Shop. He employs five barbers and pays each a base rate of $1,000 per month. One of the barbers serves as the manager and receives an extra $500 per month. In...
-
Calculate the change in entropy for the conversion of \(1 \mathrm{~mol}\) of ice to liquid at \(273 \mathrm{~K}\) and \(1 \mathrm{~atm}\). The latent heat of fusion is \(6500 \mathrm{~J} /...
-
Calculate the change in entropy when 5 moles of an ideal gas expands from a volume of \(5 \mathrm{~L}\) to \(50 \mathrm{~L}\) at \(27^{\circ} \mathrm{C}\).
-
Calculate the entropy change when \(96 \mathrm{~g}\) of methane is heated from \(35^{\circ} \mathrm{C}\) to \(200^{\circ} \mathrm{C}\) at constant volume. Assume \(C_{V}=1.735 \mathrm{~kJ} /...

Study smarter with the SolutionInn App


