Experiment #7 LIMITING REACTANT Name REPORT SHEET DATA Table A. Limiting Reactant Data Description Color of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
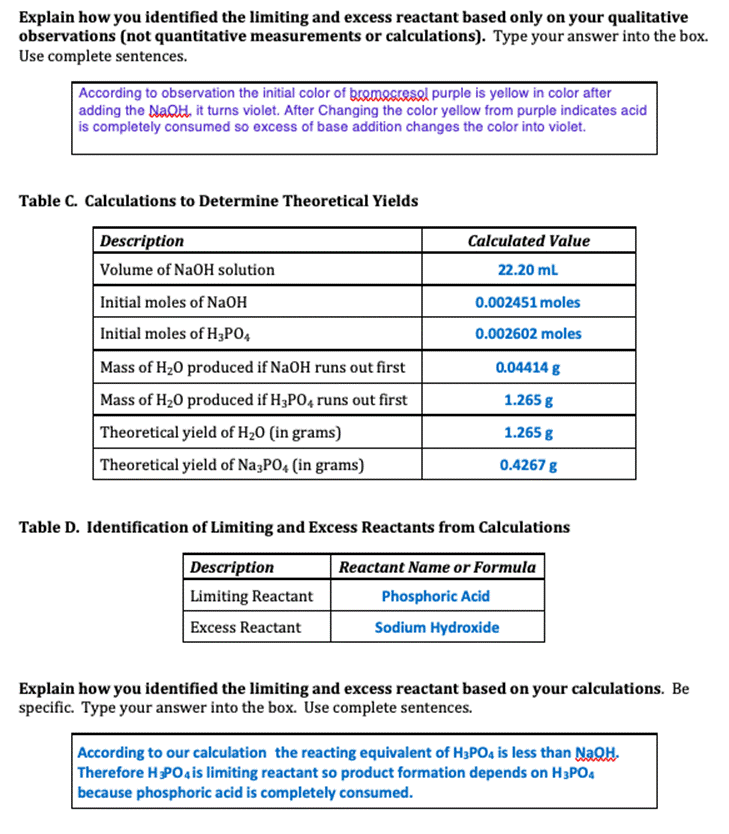
Experiment #7 LIMITING REACTANT Name REPORT SHEET DATA Table A. Limiting Reactant Data Description Color of bromocresol purple in NaOH solution Measurement or Observation Purple Color of bromocresol purple in H3PO4 solution Yellow Concentration of H3PO4 solution 0.1041 M Volume of H3P04 solution 25.00 mL Concentration of NaOH solution 0.1104 M Initial buret reading for NaOH solution 14.00 ml Final buret reading for NaOH solution 36.20 mL Color of reaction mixture after bromocresol purple added Yellow DATA ANALYSIS SUMMARY Balanced Chemical Equation for Reaction: 3NAOH + H3PO4 ------- -→ Na Po4 +3H20 Table B. Identification of Limiting and Excess Reactants from Qualitative Observations Description Reactant Name or Formula Limiting Reactant Phosphoric Acid Excess Reactant Sodium Hydroxide Explain how you identified the limiting and excess reactant based only on your qualitative observations (not quantitative measurements or calculations). Type your answer into the box. Use complete sentences. According to observation the initial color of gromocresel purple is yellow in color after adding the NaQH. it turns violet. After Changing the color yellow from purple indicates acid is completely consumed so excess of base addition changes the color into violet. Table C. Calculations to Determine Theoretical Yields Description Calculated Value Volume of NaOH solution 22.20 ml Initial moles of NaOH 0.002451 moles Initial moles of H3PO4 0.002602 moles Mass of H20 produced if NaOH runs out first 0.04414 g Mass of H20 produced if H3PO4 runs out first 1.265 g Theoretical yield of H20 (in grams) 1.265 g Theoretical yield of Na3PO4 (in grams) 0.4267 g Table D. Identification of Limiting and Excess Reactants from Calculations Description Reactant Name or Formula Limiting Reactant Phosphoric Acid Excess Reactant Sodium Hydroxide Explain how you identified the limiting and excess reactant based on your calculations. Be specific. Type your answer into the box. Use complete sentences. According to our calculation the reacting equivalent of H3PO4 is less than NaOH. Therefore HPO4 is limiting reactant so product formation depends on H3PO4 because phosphoric acid is completely consumed. Table E. Calculations to Determine Left Over Reactant Amounts Description Calculated Value Volume of water produced Total volume of solution in beaker after reaction Moles of NaOH left over in beaker Moles of H3PO4 left over in beaker Final molarity of NaOH in beaker Final molarity of H3PO4 in beaker Experiment #7 LIMITING REACTANT Name REPORT SHEET DATA Table A. Limiting Reactant Data Description Color of bromocresol purple in NaOH solution Measurement or Observation Purple Color of bromocresol purple in H3PO4 solution Yellow Concentration of H3PO4 solution 0.1041 M Volume of H3P04 solution 25.00 mL Concentration of NaOH solution 0.1104 M Initial buret reading for NaOH solution 14.00 ml Final buret reading for NaOH solution 36.20 mL Color of reaction mixture after bromocresol purple added Yellow DATA ANALYSIS SUMMARY Balanced Chemical Equation for Reaction: 3NAOH + H3PO4 ------- -→ Na Po4 +3H20 Table B. Identification of Limiting and Excess Reactants from Qualitative Observations Description Reactant Name or Formula Limiting Reactant Phosphoric Acid Excess Reactant Sodium Hydroxide Explain how you identified the limiting and excess reactant based only on your qualitative observations (not quantitative measurements or calculations). Type your answer into the box. Use complete sentences. According to observation the initial color of gromocresel purple is yellow in color after adding the NaQH. it turns violet. After Changing the color yellow from purple indicates acid is completely consumed so excess of base addition changes the color into violet. Table C. Calculations to Determine Theoretical Yields Description Calculated Value Volume of NaOH solution 22.20 ml Initial moles of NaOH 0.002451 moles Initial moles of H3PO4 0.002602 moles Mass of H20 produced if NaOH runs out first 0.04414 g Mass of H20 produced if H3PO4 runs out first 1.265 g Theoretical yield of H20 (in grams) 1.265 g Theoretical yield of Na3PO4 (in grams) 0.4267 g Table D. Identification of Limiting and Excess Reactants from Calculations Description Reactant Name or Formula Limiting Reactant Phosphoric Acid Excess Reactant Sodium Hydroxide Explain how you identified the limiting and excess reactant based on your calculations. Be specific. Type your answer into the box. Use complete sentences. According to our calculation the reacting equivalent of H3PO4 is less than NaOH. Therefore HPO4 is limiting reactant so product formation depends on H3PO4 because phosphoric acid is completely consumed. Table E. Calculations to Determine Left Over Reactant Amounts Description Calculated Value Volume of water produced Total volume of solution in beaker after reaction Moles of NaOH left over in beaker Moles of H3PO4 left over in beaker Final molarity of NaOH in beaker Final molarity of H3PO4 in beaker
Expert Answer:
Answer rating: 100% (QA)
9 Pou voleme Conc 01oUI M 25 mL 17 So mote 01041 A 24 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A quantity of 0.25 M sodium hydroxide is added to a solution containing 0.15 mol of acetic acid. The final volume of the solution is 375 mL and the pH of this solution is 4.45. a. What is the molar...
-
It takes 38 mL of 0.75 M NaOH solution to completely neutralize 155 mL of a sulfuric acid solution (H2SO4). What is the concentration of the H2SO4 solution?
-
You have 75.0 mL of 0.10 M HA. After adding 30.0 mL of 0.10 M NaOH, the pH is 5.50. What is the Ka value of HA?
-
For the following exercises, use shells to find the volume generated by rotating the regions between the given curve and y = 0 around the x-axis. 130. y = 1-x,x = 0, and x = 1 131. y = x, x = 0, and...
-
Now your boss wants you to evaluate the dividend policy of Information Systems, Inc. (ISI), which develops software for the health care industry. ISI was founded five years ago by Donald Brown and...
-
Topic is Theory Of Constraints. I think the problem is how it treats many items as fixed and inflexible. The body of the paper must be between 750 and 1000 words (approximately 3 pages) 3 credible...
-
The fiscal year of the Eddy Company ends on December 31. Your audit report, dated February 26, is to be delivered to the client on March 9. Listed below are events that occur or are discovered from...
-
Gordeeva Corporation began selling goods on the installment basis on January 1, 2012. During 2012, Gordeeva had installment sales of $150,000; cash collections of $54,000; cost of installment sales...
-
Rosie's Florist borrows $300,000 to be paid off in six years. The loan payments are semiannual with the first payment due in six months, and interest is at 6%. What is the amount of each payment? 3)...
-
Submag, Inc., uses direct-mail promotion to sell magazine subscriptions. Magazine publishers pay Submag $3.12 for each new subscription. Submags costs include the expenses of printing, addressing,...
-
Sara Nade had a 2021 capital asset transaction as noted below. For each asset disposition, determine the amount of her gain or loss for tax purposes. On February 2 nd Sara received 100 shares of Unk,...
-
Describe lump-sum or alimony in gross. How is it different from other forms of alimony in its nature and effect?
-
Jupiter, with an inertia 317.83 times that of Earth, is at an average distance of \(7.784 \times 10^{11} \mathrm{~m}\) from the Sun. At what distance from the centre of the Sun is the center of mass...
-
Define rehabilitative alimony and identify its primary purpose.
-
Etsy is an e-commerce website focusing on handmade or vintage items and unique factory-manufactured products. These items cover a wide range of product types, including jewelry, furniture,...
-
Identify the four bases of jurisdiction under the UCCJA and indicate how the UCCJEA and PKPA refine them.
-
Q2: explain the difference between the peak input voltage Vp(in) and the output half-wave Vp(rect) Q3: Explain the difference between the peak input voltage Vp(in) and the output Full-wave Vp(rec)...
-
Pappa's Appliances uses the periodic inventory system. Details regarding the inventory of appliances at January 1, purchases invoices during the year, and the inventory count at December 31 are...
-
Find a Cartesian equation of the plane through the three points (2, 3, -1), (-1, 5, 2), and (-4, -2, 2).
-
Prove that is the relative rate of change is a positive constant then the function must represent exponential growth.
-
Solve the following inequalities: (a) |x - 2| < 1 (b) |x - 2| 1 (c) |x - 2| < 0.1 (d) |x - 2| < 0.01
-
List some BOs for this term and explain their role.
-
Identify the EBT for the word Change and give reasons for this choice.
-
Create three more patterns for change. Use three different scenarios.

Study smarter with the SolutionInn App


