-Identify the moles of each reactant used. -Considering the aldol reaction and its mechanism, write a...
Fantastic news! We've Found the answer you've been seeking!
Question:
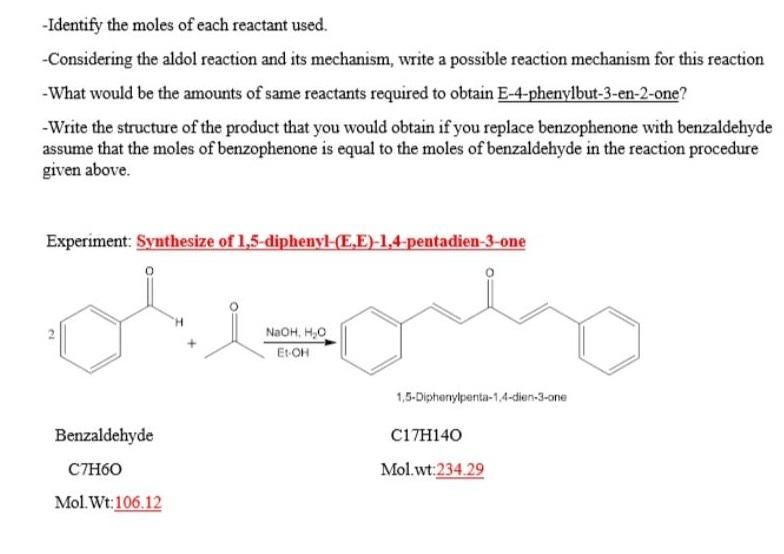
Transcribed Image Text:
-Identify the moles of each reactant used. -Considering the aldol reaction and its mechanism, write a possible reaction mechanism for this reaction -What would be the amounts of same reactants required to obtain E-4-phenylbut-3-en-2-one? -Write the structure of the product that you would obtain if you replace benzophenone with benzaldehyde assume that the moles of benzophenone is equal to the moles of benzaldehyde in the reaction procedure given above. Experiment: Synthesize of 1,5-diphenyl-(E,E)-1,4-pentadien-3-one NaOH, H,0 Et-OH 1,5-Diphenylpenta-1,4-dien-3-one Benzaldehyde C17H140 CTH60 Mol.wt:234.29 Mol. Wt:106.12 2. -Identify the moles of each reactant used. -Considering the aldol reaction and its mechanism, write a possible reaction mechanism for this reaction -What would be the amounts of same reactants required to obtain E-4-phenylbut-3-en-2-one? -Write the structure of the product that you would obtain if you replace benzophenone with benzaldehyde assume that the moles of benzophenone is equal to the moles of benzaldehyde in the reaction procedure given above. Experiment: Synthesize of 1,5-diphenyl-(E,E)-1,4-pentadien-3-one NaOH, H,0 Et-OH 1,5-Diphenylpenta-1,4-dien-3-one Benzaldehyde C17H140 CTH60 Mol.wt:234.29 Mol. Wt:106.12 2.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In the diet example in this chapter, what would be the effect on the optimal solution of increasing the minimum calorie requirement for the breakfast to 500 calories? to 600 calories? Increase the...
-
1). A firm has entered into a 4-year, annual-pay, 6% plain vanilla interest rate swap with a notional principal value of $10,000,000. The firm is the fixed rate payer (i.e. the swap dealer is the...
-
A possible mechanism for a gas-phase reaction is given below. What is the rate law predicted by this mechanism? (fast equilibrium) 2NOCI NOC NO2NOCI (slow)
-
Yamini and Fatima, two students of Class IX of a school, together contributed Rs. 100 towards the Prime Ministers Relief Fund to help the earthquake victims. Write a linear equation which satisfies...
-
A firm is currently employing an "aggressive" working capital policy with regard to the level of current assets it maintains (relatively low levels of current assets for each possible level of...
-
During a marathon race, a runners blood flow increases to 10.0 times her resting rate. Her bloods viscosity has dropped to 95.0% of its normal value, and the blood pressure difference across the...
-
There are several exceptions to the hearsay rule. Identify at least three exceptions and give an example of each.
-
1. Offender is at least 18. 2. Offender is white. 3. Offender is male. 4. Victim is a white female. 5. Victim is either white or female. 6. Victim and offender are from the same age category. 7....
-
explain the penalties that are given if they fail to achieve the targets in a ladies medical center?
-
A. Richard McCarthy (born 2/14/64; Social Security number 100-10-9090) and Christine McCarthy (born 6/1/1966; Social security number 101-21-3434) have a 19-year-old son (born 10/2/99 Social Security...
-
The artical 2020 Construction Trends: 30+ Experts Weigh In https://constructionblog.autodesk.com/2020-construction-trends/ Newsday Grading Rubric Introduction to Public Speaking Read this note...
-
Why should the litigation crisis facing auditors be of concern to other businesspeople, professionals, senators, or others?
-
Identify several proposed legal reforms and state how they would contribute to resolving the litigation crisis.
-
Describe the basic operation of a floating gate MOSFET and how this can be used in an erasable ROM.
-
a. Identify three types of substantive tests and indicate how they relate to one another. b. What is a dual-purpose test?
-
a. What is the overall objective of a financial statement audit? b. How does the auditor customarily meet this overall objective?
-
In the 2016 Olympic games, Deng Wei set a women's record of 147 kg in the clean and jerk. Suppose that she lifted 135 kg during practice. How much work W did she do on the weights to lift them above...
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
A California grower has a 50-acre farm on which to plant strawberries and tomatoes. The grower has available 300 hours of labor per week and 800 tons of fertilizer, and he has contracted for shipping...
-
In Problem 15, Computers Unlimited wants to better meet demand at the four universities it supplies. It is considering two alternatives: (1) Expand its warehouse at Richmond to a capacity of 600, at...
-
In Problem 53, the decision analysis is for automobiles and light trucks. Blue Ridge Power and Light would like to reformulate the problem for its heavy construction equipment. Emergency maintenance...
-
Is there or can there be an end to digital business transformation?
-
Could an accountant work at a more mature digital business like Google or Amazon without digital technology / digital transformation / digital business competencies?
-
How do digital technology advancements disrupt the work accountants do?

Study smarter with the SolutionInn App



