Explain why the first ionization energy of Ca is greater than that of K, whereas the...
Fantastic news! We've Found the answer you've been seeking!
Question:
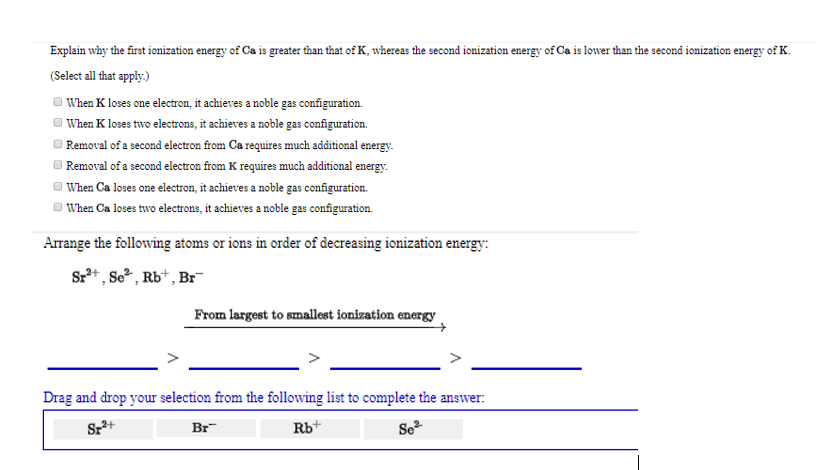
Transcribed Image Text:
Explain why the first ionization energy of Ca is greater than that of K, whereas the second ionization energy of Ca is lower than the second ionization energy of K. (Select all that apply.) When K loses one electron, it achieves a noble gas configuration. When K loses two electrons, it achieves a noble gas configuration. Removal of a second electron from Ca requires much additional energy. Removal of a second electron from K requires much additional energy. When Ca loses one electron, it achieves a noble gas configuration. When Ca loses two electrons, it achieves a noble gas configuration. Arrange the following atoms or ions in order of decreasing ionization energy: Sr²+, So², Rb+, Br From largest to smallest ionization energy Drag and drop your selection from the following list to complete the answer: Sz²+ Br Rb+ Se² Explain why the first ionization energy of Ca is greater than that of K, whereas the second ionization energy of Ca is lower than the second ionization energy of K. (Select all that apply.) When K loses one electron, it achieves a noble gas configuration. When K loses two electrons, it achieves a noble gas configuration. Removal of a second electron from Ca requires much additional energy. Removal of a second electron from K requires much additional energy. When Ca loses one electron, it achieves a noble gas configuration. When Ca loses two electrons, it achieves a noble gas configuration. Arrange the following atoms or ions in order of decreasing ionization energy: Sr²+, So², Rb+, Br From largest to smallest ionization energy Drag and drop your selection from the following list to complete the answer: Sz²+ Br Rb+ Se²
Expert Answer:
Answer rating: 100% (QA)
Answer 1 The first ionization energy of an element is the energy required to remove one electron from a neutral atom of that element Calcium has an at... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Explain why the first ionization energy tends to increase as one proceeds from left to right across a period. Why is the first ionization energy of aluminum lower than that of magnesium and the first...
-
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008...
-
From Figure 8.18, predict the first ionization energy of francium (Z = 87). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008 At...
-
Ebrahim Patel is a wholesaler who uses the periodic inventory system to account for inventory. Transactions for February: 1 Bought inventory from Rich Traders for R5 000 on credit. 2 Sold inventory...
-
What are the potential causes of action against an auditor under a breach of contract lawsuit?
-
Calculate the transconductance of an n-channel MOSFET at I D = 10 nA and V DS = 1 V, assuming subthreshold operation and n = 1.5. Assuming (C gs + C gd + C gb ) = 10 fF, calculate the corresponding...
-
A Gallup poll taken in May 2016 asked 1025 adult Americans to state their opinion on the availability of abortions. The following table presents the results, along with the proportions of people who...
-
Joint-cost allocation. Elsie Dairy Products Corp buys one input full-cream milk, and refines it in a churning process. From each gallon of milk Elsie produces two cups (one pound) of butter and two...
-
Cheyenne Manufacturing Corporation uses both standards and budgets. The company estimates that production for the year will be 100,000 units of Product Fast. To produce these units of Product Fast,...
-
a. Tables 1 through 5 contain the financial information describing the effects of level production on inventory, cash flow, loan balances, and interest expense. Reproduce these tables as if Tim's...
-
Choose 2 accounts on your financial statements to write up an audit program, i.e. the substantive procedures, to collect the evidence to audit your accounts. You should clearly identify each step...
-
Would you want to live in a society where crime rates were quite low because they were controlled by extremely harsh punishment, such as flogging for vandalism? Are criminals rational decision...
-
The parents stay for a while and play with their child and the other children and seem happy to leave. They say goodbye and their child cries initially when they leave and needs comforting. You sit...
-
Find the compound interest and future value. Round your answers to the nearest cent. Do not round intermediate steps. Principal Rate Compounded $760 5% Daily Time 1 year The future value is and the...
-
Is the term (x+2) a factor of the polynomial shown below? f(x)=x-10x+27x-12
-
What do you think...How has the innovation discussed above influenced our lives and the way we interact with each other? Discuss your thoughts on some of the relational topics listed below. Support...
-
The Case GoNative Pty. Ltd. is based in Melbourne, Victoria. The business was established in 2015 to cater for the demand for Australian native food products. The company manufactures and retails a...
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
Draw cyclic structures for D-ribose and D-mannose.
-
Balance the following equations. a. Cr(s) + S8(s) ( Cr2S3(s) Heat b. NaHCO3(s) ( Na2CO3(s) + CO2(g) + H2O(g) Heat c. KClO3(s) ( KCl(s) + O2(g) d. Eu(s) + HF(g) ( EuF3(s) + H2(g) e. C6H6(l) + O2(g) (...
-
What is the valence electron configuration for alkaline earth metals? List some common properties of alkaline earth metals. How are alkaline earth metals prepared?
-
Applying the criterion for equilibrium, derive the Clausius-Clapeyron equation.
-
A binary liquid mixture consists of \(60 \mathrm{~mol}\) per cent ethylene and \(40 \mathrm{~mol}\) per cent propylene. At \(423 \mathrm{~K}\), the vapour pressure of ethylene and propylene are...
-
The pure component vapour pressure of two organic liquids \(\mathrm{X}\) and \(\mathrm{Y}\) by Antoine equations are given by \[ \ln P_{1}^{\text {Sat }}=14.35-\frac{2942}{T+220} \] and \[...

Study smarter with the SolutionInn App


