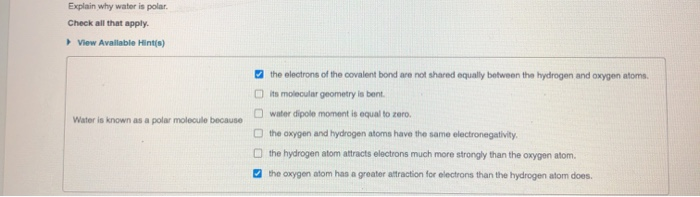
Explain why water is polar. Check all that apply. View Available Hint(s) Water is known as...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Explain why water is polar. Check all that apply. View Available Hint(s) Water is known as a polar molecule because the electrons of the covalent bond are not shared equally between the hydrogen and oxygen atoms. its molecular geometry is bent. water dipole moment is equal to zero. the oxygen and hydrogen atoms have the same electronegativity. the hydrogen atom attracts electrons much more strongly than the oxygen atom. the oxygen atom has a greater attraction for electrons than the hydrogen atom does. Explain why water is polar. Check all that apply. View Available Hint(s) Water is known as a polar molecule because the electrons of the covalent bond are not shared equally between the hydrogen and oxygen atoms. its molecular geometry is bent. water dipole moment is equal to zero. the oxygen and hydrogen atoms have the same electronegativity. the hydrogen atom attracts electrons much more strongly than the oxygen atom. the oxygen atom has a greater attraction for electrons than the hydrogen atom does.
Expert Answer:
Answer rating: 100% (QA)
Water is a polar molecule because it has a net dipole moment Wate... View the full answer

Related Book For 

Basic Marketing Research
ISBN: 978-1133188544
8th edition
Authors: Tom J. Brown, Tracy A. Suter, Gilbert A. Churchill
Posted Date:
Students also viewed these chemistry questions
-
Check All That Apply Mass customization strategies can be applied to all products. The key to mass customization is postponement Postponement depends upon the use of standardization and...
-
a. How many hydrogen and oxygen atoms are present in 1 molecule of H2O? b. How many moles of hydrogen and oxygen atoms are present in 1 mol H2O? c. What are the masses of hydrogen and oxygen in 1.0...
-
Explain why water is more effective than lead in slowing down fast neutrons.
-
In which control account would you expect to find a provision for doubtful debts?
-
Consider the situation given in Example 17.3. After the Series C investment by Owl, is the Series A implied LP valuation lower than its original LP cost?
-
True or False? Discuss. a. In a progressive tax rate system, risk neutral investors prefer volatile assets over riskless assets because they can average their tax rates. b. In a progressive tax rate...
-
General Manufacturing Company makes residential aluminum windows. A company has offered to supply General with the window crank assembly it needs for $3.50 each. General uses 50,000 crank assemblies...
-
Spear Custom Furniture uses an activity-based cost accounting system to apply overhead to production. The company maintains four overhead cost pools. The four cost pools, and their budgeted amounts...
-
Question No. 2 Accounting Changes Homer Alaska Inc. (HA) has used the completed-contract method to account for long-term construction contracts since its inception in 2018. In 2020, HA decided to...
-
Best Buy Co., Inc. is a leading provider of technology products. Customers can shop at more than 1,700 stores or online. The company is also known for its Geek Squad for technology services. Suppose...
-
"A company is responsible for ensuring timely action oncommunicating delays or changes to flights/cruises/bus transportwith their passengers. This company currently uses traditionalexcel database t 2...
-
Name two relationships between auditors and clients that are prohibited by the AICPA because independence will be impaired.
-
Name and briefly describe the purpose of each of the eight sections of the Principles of Professional Conduct.
-
Googles dominance in search and other online spaces allows it to collect vast amounts of data that help it deliver targeted advertising. In fact, advertising revenues from the Google unit accounted...
-
What is the basic structure of the Code of Professional Conduct?
-
What was the purpose of the Anderson Report?
-
Ethical and Cultural Considerations in Groups Ethical considerations are important to planning the structure of any group and will be a key component of the group proposal that will come together in...
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
Suppose you were asked to design an appropriate communication method to find out students' feelings and opinions about the various food services available on campus. a. What degree of structure would...
-
Using the steps of the research process to structure your thinking, evaluate the following marketing research effort: The FlyRight Airline Company was interested in altering the interior layout of...
-
A retailer of household appliances is planning to introduce a new brand of dishwashers to the local market and wishes to estimate demand for the product. He has decided to use two stage area sampling...
-
Implement the Gibbs sampler for the mixture model. Compute \(\hat{R}\) and the autocorrelation of the component means \(\mu_{k}\). How many samples are required for convergence? How much should the...
-
Marginalise the Gaussian mean from the Gibbs sampler to give a conditional distribution for \(z_{n k}\) conditioned only on the other assignments and the prior parameters \(\left(\mu_{0},...
-
Implement the collapsed Gibbs sampler for the Gaussian mixture model. At each iteration in the sampler, sample the means \(\mu_{k}\) even though they are not needed (sample them from the posterior...

Study smarter with the SolutionInn App


