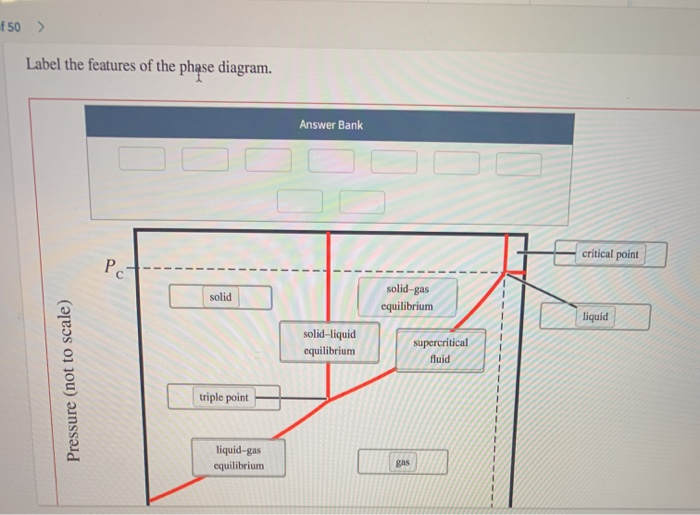
f 50 > Label the features of the phase diagram. Pressure (not to scale) Pc- solid...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
f 50 > Label the features of the phase diagram. Pressure (not to scale) Pc- solid triple point liquid-gas equilibrium Answer Bank solid-liquid equilibrium solid-gas equilibrium gas supercritical fluid critical point liquid f 50 > Label the features of the phase diagram. Pressure (not to scale) Pc- solid triple point liquid-gas equilibrium Answer Bank solid-liquid equilibrium solid-gas equilibrium gas supercritical fluid critical point liquid
Expert Answer:
Answer rating: 100% (QA)
The lebels are correctly placed in the phase diagram of CO as follows Pressure not to scal... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The pressure of a gas at the triple point of water is 1.35 atm. If its volume remains unchanged, what will its pressure be at the temperature at which CO2 solidifies?
-
Label the regions of the phase diagram in Fig. 6.3 7. State what substances (if compounds give their formulas) exist in each region. Label each substance in each region as solid, liquid, or gas.
-
The equilibrium pressure of H, over solid uranium and uranium hydride, UH3' at 500 K is 139 Pa. Calculate the standard Gibbs energy of formation ofUH3 (s) at 500 K.
-
Maicom Construction Materials Inc. , hereinafter referred to as "MCM", is a construction materials company established in Moncton, New Brunswick. Its facilities (warehouse, store and offices) are...
-
Using data from the CVS Corporation annual report in the Supplement to Chapter 5, conduct a comprehensive ratio analysis that compares the companys performance in 2008 and 2007. If you have computed...
-
A control system has two forward paths, as shown in Figure P4.4. (a) Determine the overall transfer function T(s) = Y(s)/R(s). (b) Calculate the sensitivity, STG, using Equation (4.16). (c) Does the...
-
Consider the set of vectors \((-1,1,1),(1,-1,1),(1,1,-1)\). a. Use the Gram-Schmidt process to find an orthonormal basis for \(R^{3}\) using this set in the given order. b. What do you get if you do...
-
Powell Company produces a single product. Its income statement under absorption costing for its first two years of operation follow. Additional Information a. Sales and production data for these...
-
Question 18 1.5 / 1.5 pts Dave's Burgers began the year with total assets of $420,000 and shareholders' equity of $82,500. During the year, the company earned $100,000 in net income and paid $35,000...
-
A storage tank is connected to a pond (at atmospheric pressure!) by a length of 3-in pipe and two gate valves. From previous operating experience, it has been found that when the tank is at a...
-
Java Assume the availability of class named DateManager that provides a static method , printTodaysDate, that accepts no arguments and returns no value. Write a statement that calls printTodaysDate.
-
When you added up the value of these clothing inventory items they came to a grand total $ 5 7 5 in estimated fair value as of 9 / 1 / 2 0 2 3 . You originally paid $ 1 , 9 7 6 for these items. You...
-
An acorn is thrown horizontally to the left by a squirrel. If the distance covered by the acorn is 5 m to the left and it took 3 s to cover that distance, determine the velocity of the acorn.
-
Robinson Products Company has two service departments (S1 and S2) and two production departments (P1 and P2). The distribution of each service department's efforts (in percentages) to the other...
-
What will balance scorecard for university operation look like when the university goal is "to increase the number of international students"? Please provide the a few example of objectives for each...
-
Please submit your work as a Word document labeled CS341T_U3_YourName.docx. Click the Edit button within the submission lesson to access the submission area. Click the Select File button to upload...
-
By-hand, calculate the z-score for Question 1_C to show why your answers to Question Sets 1_B and 1_D are the same. Show your work.
-
Players A, B, and C toss a fair coin in order. The first to throw a head wins. What are their respective chances of winning?
-
The oxides of nitrogen are very important components in urban air pollution. Name each of the following compounds: (a) N2O (b) NO (c) NO2 (d) N2O5 (e) N2O4.
-
Acetylene gas, C2H2 (g), can be prepared by the reaction of calcium carbide with water: CaC2 (s) + 2 H2O (l) Ca (OH)2 (s) + C2H2 (g) Calculate the volume of C2H2 that is collected over water at by...
-
A reaction that contributes to the depletion of ozone in the stratosphere is the direct reaction of oxygen atoms with ozone: At 298 K the rate constant for this reaction is 4.8 Ã 105 M-1 s-1....
-
Another population model is one in which species compete for resources, such as a limited food supply. Such a model is given by \[\begin{aligned} & x^{\prime}=a x-b x^{2}-c x y \\ & y^{\prime}=d y-e...
-
Consider the period of a nonlinear pendulum. Let the length be \(L=1.0\) \(\mathrm{m}\) and \(g=9.8 \mathrm{~m} / \mathrm{s}^{2}\). Sketch \(T\) versus the initial angle \(\theta_{0}\), and compare...
-
Derive the first integral of the Lotka-Volterra system, \(a \ln y+d \ln x-\) \(c x-b y=C\).

Study smarter with the SolutionInn App


