f(n)= n? / log n and g(n) = n(log n)? Select one: O a. f=(g) O...
Fantastic news! We've Found the answer you've been seeking!
Question:
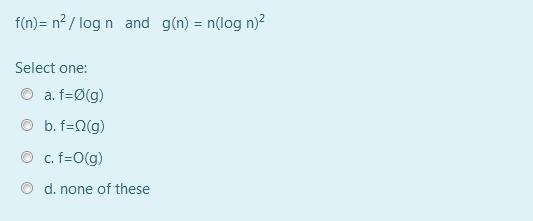
Transcribed Image Text:
f(n)= n? / log n and g(n) = n(log n)? Select one: O a. f=Ø(g) O b. f=N(g) O c. f=O(g) d. none of these f(n)= n? / log n and g(n) = n(log n)? Select one: O a. f=Ø(g) O b. f=N(g) O c. f=O(g) d. none of these
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these algorithms questions
-
In linear programming, what-if analysis is associated with determining the effect of changing: I. Objective function coefficients II. Right-hand side values of constraints III. Decision variable...
-
Let S = {vi, o,., Tm} C R". WVhich of the following is correct. .... (a) If m < n, then S must be linearly independent (b) If m < n, then S must be linearly dependent (c) If m > n, then S must be...
-
On January 1. Ruiz Company issued bonds as follows: Face Value: Number of Years: Stated Interest Rate: Interest payments per year 500,000 15 7% Required: 1) Calculate the bond selling price given the...
-
What is the charge of the iron ion released upon dissolvingFe(NH 4 ) 2 (SO 4 ) 2 ?Remember the charges of the polyatomic ions NH 4 andSO 4 and that the entire compound will be chargeneutral. a). 8+...
-
Reexamine the capital investment decision in the disposable diaper industry (Example 15.4) from the point of view of an incumbent firm. If P&G or Kimberly-Clark were to expand capacity by building...
-
Assume the same probabilities as in the previous question. For a randomly selected family with three children, whats the probability of (a) Three boys? (b) Three girls? (c) Either three boys or three...
-
The L-shaped frame is made from two segments, each of length \(L\) and flexural stiffness \(E I\). Determine the horizontal displacement of point \(C\). W C L A B- -L-
-
Income Statement for a Merchandiser Fill in the missing amounts in the following income statement for Carpenters Department Store Inc. $125,600 (al Less: Sales returns and allowances $122,040 $23,400...
-
Icebreaker Company (a U.S.-based company) purchases materials from a foreign supplier on December 1, 2020, with payment of 26,000 dinars to be made on March 1, 2021. The materials are consumed...
-
The following transactions of Beltville College transpired during 2020. The funds necessary are the Endowment Fund, the Annuity Fund, the Plant Fund?Unexpended, the Plant Fund?Investment in Plant,...
-
Assume that Evergreen Healthcare, a provider of skilled nursing facility services, is evaluating the feasibility of building a new facility to replace one of its aging facilities in a small,...
-
Prescott Inc. sells coffee mugs and water bottles. Sales of coffee mugs traditionally run around 2 7 5 , 0 0 0 per year and sales of water bottles are 5 0 0 , 0 0 0 sharply increasing over the past 3...
-
On November 1 , the company agreed to do a special six - month course for a client. The contract calls for a monthly fee of $ 9 0 0 , and the client paid the first five months' revenue in advance....
-
2 Required: 1. Using the company's plantwide approach: a. Compute the plantwide predetermined rate for the current year. b. Determine the amount of manufacturing overhead cost that would have been...
-
Problem 2 - Multiple Production Department Factory Overhead Rates The total factory overhead for Bardot Marine Company is budgeted for the year at $471,750 divided into two departments: Fabrication,...
-
Arrow Construction Company Ltd . purchased a farm from K . Jones. Arrow and Jones completed the transaction under the following terms: a check from Arrow to Jones for $ 1 4 0 , 0 0 0 ; bank loan...
-
A company is adding an additional machine to increase its operating capacity. As utilization of the new machine ramps up, the company will see changes in revenues, operating costs, and taxes. For a...
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
Compare the half-life equations for a first-order and a second-order reaction. For which reaction order is the value of the half-life independent of the reactant concentration?
-
At what speed must an electron travel to have a wavelength of 10.0 pm?
-
Suppose you need to boil a water-based solution at a temperature lower than 100C. What kind of liquid could you add to the water to make this happen?
-
The nozzle of a mixing vibrator is tested for its number of vibrations. The vibration frequency, for each nozzle sample, can be modeled by a normal distribution with mean 128 and standard deviation...
-
If a random variable has the standard normal distribution, find the probability that it will take on a value (a) between 0 and 2.3 ; (b) between 1.22 and 2.43 ; (c) between -1.45 and -0.45 ; (d)...
-
Verify that (a) \(z_{0.005}=2.575\); (b) \(z_{0.025}=1.96\).

Study smarter with the SolutionInn App


