Following is a partial production cost report for Mitchell Manufacturing's Canning department. Equivalent units of production...
Fantastic news! We've Found the answer you've been seeking!
Question:
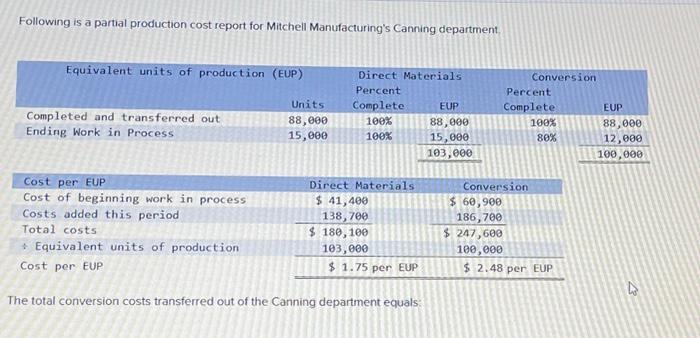
Transcribed Image Text:
Following is a partial production cost report for Mitchell Manufacturing's Canning department. Equivalent units of production (EUP) Direct Materials Percent Conversion Units Complete EUP Percent Complete EUP Completed and transferred out Ending Work in Process 88,000 100% 15,000 100% 88,000 15,000 100% 88,000 80% 12,000 103,000 100,000 Cost per EUP Cost of beginning work in process Costs added this period Total costs Equivalent units of production Cost per EUP $ 41,400 138,700 $ 180,100 103,000 $ 1.75 per EUP The total conversion costs transferred out of the Canning department equals: Direct Materials Conversion $ 60,900 186,700 $ 247,600 100,000 $ 2.48 per EUP 13 Following is a partial production cost report for Mitchell Manufacturing's Canning department. Equivalent units of production (EUP) Direct Materials Percent Conversion Units Complete EUP Percent Complete EUP Completed and transferred out Ending Work in Process 88,000 100% 15,000 100% 88,000 15,000 100% 88,000 80% 12,000 103,000 100,000 Cost per EUP Cost of beginning work in process Costs added this period Total costs Equivalent units of production Cost per EUP $ 41,400 138,700 $ 180,100 103,000 $ 1.75 per EUP The total conversion costs transferred out of the Canning department equals: Direct Materials Conversion $ 60,900 186,700 $ 247,600 100,000 $ 2.48 per EUP 13
Expert Answer:
Answer rating: 100% (QA)
To determine the total conversion costs transferred out of the ... View the full answer

Posted Date:
Students also viewed these accounting questions
-
The number of claims filed each week with Security Insurance Company has a mean of 700 and a standard deviation of 250. Calculate the probability that the number of claims this week will be: (a)...
-
In the United States, consumption taxes (such as sales taxes) are typically state rather than federal taxes. In Europe, consumption taxes are imposed nationally rather than regionally, and they are...
-
A branch current is described by Determine: (a) The characteristic equation, (b) The type of damping exhibited by the circuit, (c) i(t) given that i(0) 1 and di(0) / dt = 2 . +10itt) 0 dt dt
-
What is the As-Maintained Configuration, and when is it established?
-
Condensed balance sheet and income statement data for Glassgow Corporation are presented here. Additional information:1. The market price of Glassgow's common stock was $7.00, $7.50, and $8.50 for...
-
On January 1, 20X1, ABC Company purchased a machine for use in its factory. ABC started using the machine on February 1, 20X1. During 20X1, ABC spent the following on the machine and on expenditures...
-
1. Smith Corp. stock had the following information: Price as of 12/31/19 $75 per share Price as of 12/31/18 $87 per share Dividend received during the year $2,50 What is the stock's total return for...
-
From the practice rounds to the end of the competition rounds in the Capsim business simulation, please provide an overall assessment of your team's performance. What were your team's strengths?...
-
I am the owner of a real estate development/home construction company. Identify and apply an appropriate analytical tool to analyze external factors that affect company performance. Given the...
-
Do you think it is right for organizations to set up organizational display rules? (1) state your position (Yes or No) and (2) give 3 reasons to justify your position. (Hint: Emotional labor)
-
Describe a strategic goal that could support the mission statement: " To provide a high-quality, low-cost product to our customers." Who would be responsible for this goal, and what kind of power...
-
Define the service package of the local hospital or individual doctor's office you are familiar with. What is its strongest element? What is its weakest one? Why? How can/should it be improved? To...
-
Read the Poem: Journey of the Magi and write a paper about it
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
The freezing point of pure benzene is \(5.44^{\circ} \mathrm{C}\) and that of the solution containing \(2.092 \mathrm{~g}\) of benzaldehyde in \(100 \mathrm{~g}\) of benzene is \(4.44^{\circ}...
-
The molality of dissolved gases in water at \(0^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\) is \(1.29 \times 10^{-3}\). The decrease in volume during melting of ice is \(0.0907 \mathrm{cc} /...
-
Calculate the mass of methyl alcohol which, when dissolved in \(100 \mathrm{~g}\) of water, would just prevent the formation of ice at \(-10^{\circ} \mathrm{C}\), given that \(K_{\mathrm{f}}\) is...

Study smarter with the SolutionInn App

