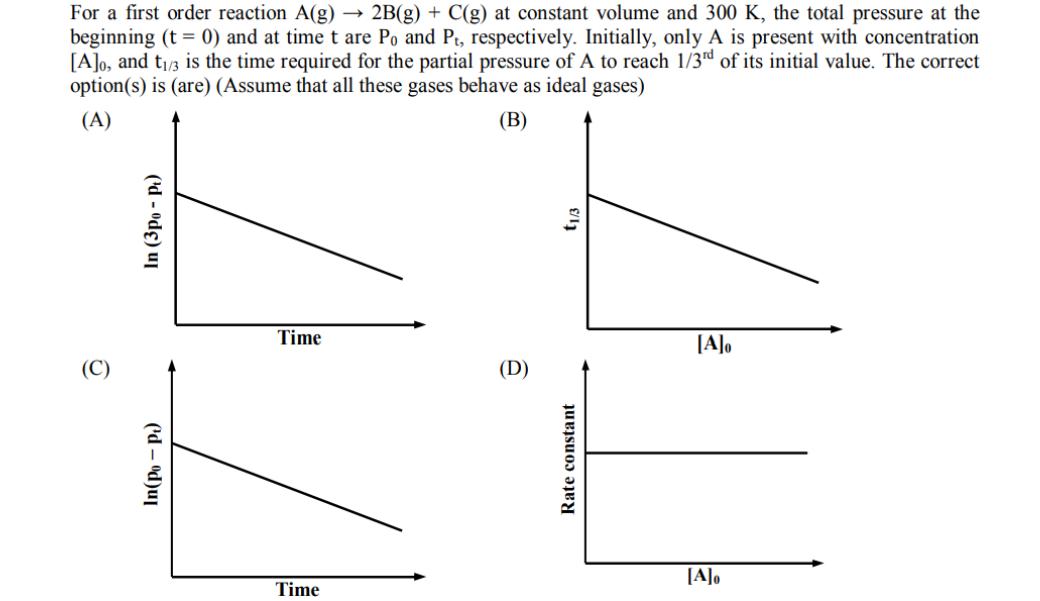
For a first order reaction A(g) 2B(g) + C(g) at constant volume and 300 K, the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For a first order reaction A(g) 2B(g) + C(g) at constant volume and 300 K, the total pressure at the beginning (t = 0) and at time t are Po and Pt, respectively. Initially, only A is present with concentration [A]o, and t/3 is the time required for the partial pressure of A to reach 1/3rd of its initial value. The correct option(s) is (are) (Assume that all these gases behave as ideal gases) (B) (A) In (3po- pt) (d-od)ul Time Time (D) t1/3 Rate constant [A]o [A]o For a first order reaction A(g) 2B(g) + C(g) at constant volume and 300 K, the total pressure at the beginning (t = 0) and at time t are Po and Pt, respectively. Initially, only A is present with concentration [A]o, and t/3 is the time required for the partial pressure of A to reach 1/3rd of its initial value. The correct option(s) is (are) (Assume that all these gases behave as ideal gases) (B) (A) In (3po- pt) (d-od)ul Time Time (D) t1/3 Rate constant [A]o [A]o
Expert Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemistry questions
-
A gaseous reaction takes place at constant volume and constant pressure in a cylinder shown here. Which of the following equations best describes the reaction? The initial temperature (T1) is twice...
-
A first order reaction begins with 176 g of reactant. After 2.6 days the amount of reactant remaining is 88 g. How much reactant will remain after 5.2 days?
-
A first order reaction requires 12 months for the concentration of the reactant to be reduced to 25% its original value. Its half life is : A) 6 months B) 12 months C) 18 months D) 24 months
-
Find the inverse of each of the given matrices by the method of Example 1 of this section. Data from Example 1 Find the inverse of the matrix First, we interchange the elements on the principal...
-
General Bearings Company is considering an investment in equipment that will replace direct labor. The equipment has a cost of $94,000, with a $6,000 residual value and an 8-year life. The equipment...
-
The band director at a high school wants to estimate the percentage of parents who support a decrease in the budget for fine arts. Because many parents attend the schools annual musical, the director...
-
It can be shown that for any three events \(A, B\), and \(C\), the probability that at least one of them will occur is given by \[\begin{aligned} P(A \cup B \cup C)= & P(A)+P(B)+P(C) \\ & -P(A \cap...
-
What is your opinion of Gap Internationals having a code of conduct for its suppliers? What would Milton Friedman say? Contrast his view with Archie Carrolls view.
-
Audit reporting processses inclusive of "what the report would be against and to whom the report may be accessible to by what methods" and rmore clarification about audit reporting ?
-
Was it absolutely necessary to "throw out" the Articles of Confederation and replace our country's constitution with the United States Constitution (USC)? Was it right to adopt the United States...
-
What is the approximate 95% prediction interval for the dependent variable when the independent variable value is 20, assuming the fitted regression line is: Y = 1.50 + 6.0(X). Assume the sample size...
-
Sandrine Machinery is a Swiss multinational manufacturing company. Currently, Sandrines financial planners are considering undertaking a 1-year project in the United States. The projects expected...
-
Nathan recently interviewed with one of the accounting firms in the city where he wants to live. The firm agreed to cover the expense of a rental car that he used to travel from his university to the...
-
Suppose you buy a December futures contract on a hypothetical 10-year, 6% semiannual coupon note with a settlement price today of 125-060. You post the initial margin required for this transaction...
-
What is the value of a call option with these data: P 5 $25, X = $25, r RF = 8%, t = 0.5 (6 months), 2 = 0.09, N(d 1 ) = 0.61586, and N(d 2 ) = 0.53287
-
Firm B has a 12% ROE. Other things held constant, what would its expected growth rate be if it paid out 25% of its earnings as dividends? 75%?
-
Leynes and Morayta form a partnership with investments of P4,000,000 and P2,000,000, respectively. Journalize their participation in net income of P3,000,000, under each of the following assumptions:...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
A hot-water pipe at 80C is losing heat to the surrounding air at 5C at a rate of 2200 W. Determine the rate of entropy generation in the surrounding air, in W/K.
-
Express Dalton's law of additive pressures. Does this law hold exactly for ideal-gas mixtures? How about nonideal-gas mixtures?
-
Argon is compressed in a polytropic process with n 5 1.2 from 120 kPa and 10oC to 800 kPa in a piston - cylinder device. Determine the work produced and heat transferred during this compression...
-
An old-fashioned tire swing exerts a force on the branch and a torque about the point where the branch meets the trunk. If you hang the swing closer to the trunk, this will --the force and-- the...
-
Which of these objects is in static equilibrium? A. B. C. D.
-
A beam with a pivot on its le ft end is suspended from a rope. Tn which direction is the force of the pivot on the beam? A. B. C. D. E.

Study smarter with the SolutionInn App



