For a metal, the electron density is 6.41028 m3. The Fermi energy is (h=6.626x10-34 Js, m...
Fantastic news! We've Found the answer you've been seeking!
Question:
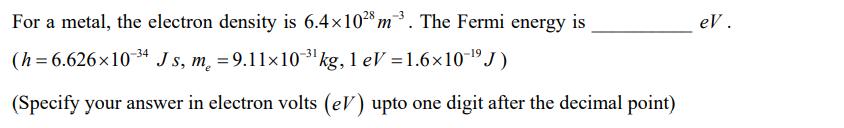
Transcribed Image Text:
For a metal, the electron density is 6.4×1028 m3. The Fermi energy is (h=6.626x10-34 Js, m = 9.11×10-3¹ kg, 1 eV = 1.6×10-¹⁹ J) (Specify your answer in electron volts (eV) upto one digit after the decimal point) eV. For a metal, the electron density is 6.4×1028 m3. The Fermi energy is (h=6.626x10-34 Js, m = 9.11×10-3¹ kg, 1 eV = 1.6×10-¹⁹ J) (Specify your answer in electron volts (eV) upto one digit after the decimal point) eV.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these physics questions
-
The Fermi energy of aluminum is 11.6 eV its density and molar mass are 2.70 g/cm3 and 27.0 g/mol, respectively. From these data, determine the number of conduction electrons per atom.
-
For a solid metal having a Fermi energy of 8.500eV, what is the probability, at room temperature, that a state having an energy of 8.520eV is occupied by an electron?
-
A 1 m3 container is filled with 400 kg of granite stone, 200 kg dry sand and 0.2 m3 of liquid 25C water. Use properties from tables A.3 and A.4. Find the average specific volume and density of the...
-
Consider constant marginal abatement cost for two firms: (a) Determine the optimal allocation for both firms when the damage function is convex, and when it is linear. (b) Is it possible to achieve...
-
During the year, Summit produces 40,000 snow shovels and sells 37,000 snow shovels. Required What is net income using full costing?
-
An asteroid is in an elliptical orbit about the sun such that its perihelion distance is d. If the eccentricity of the orbit is e, determine the aphelion distance of the orbit. Given: d = 9.30 x 10...
-
A sample of argon gas has a volume of \(0.43 \mathrm{ml}\) at 297 K. At what temperature will it have a volume of \(0.57 \mathrm{ml}\) ? (a) \(394 \mathrm{~K}\) (b) \(294 \mathrm{~K}\) (c) \(494...
-
A vending machine is programmed to count out the correct change for each transaction. Formulate and solve an IP model that will determine how change is to be made for a purchase of $4.43, when a $10...
-
i need help with this question with an explanation thanks! 7) Large Co. pays its employees $20,000 gross salary each week. 30% of salary is withheld for income taxes and 5% is withheld to pay Social...
-
Pritano Company acquired all the net assets of Succo Company on December 31, 2018, for $2,160,000 cash. The balance sheet of Succo Company immediately prior to the acquisition showed: As part of the...
-
MR PRICE CASE STUDY Mr Price: resilient and determined to become the most valuable retailer in Africa Like many retailers in South Africa, Mr Price experienced devastating civil unrest in July 2021...
-
Cullumber Company traded machinery with a book value of $ 4 9 0 0 0 0 and a fair value of $ 1 0 6 0 0 0 0 . It received in exchange from Vaughn Manufacturing a machine with a fair value of $ 9 5 4 0...
-
4. Consider the IVP t y +1 y(0) = yo- (a) Without solving, determine all values of yo for which the IVP is certain to have a unique solution. (b) Solve the IVP for all such values of yo and detemine...
-
Northwood Company manufactures basketballs. The company has a ball that sells for $ 2 5 . At present, the ball is manufactured in a small plant that relies heavily on direct labor workers. Thus,...
-
At the beginning of 2 0 2 4 , VHF Industries acquired a machine with a fair value of $ 6 , 0 7 4 , 7 0 0 by signing a four - year lease. The lease is payable in four annual payments of $ 2 million at...
-
Suppose a couple in their early 30s is planning to buy a new HDB flat. The selling price of the HDB flat is $350,000. The couple has sufficient cash to make a down-payment of $35,000 (from CPF and...
-
Porter has argued that there are four main routes to gaining competitive advantage which he calls Generic Strategies. a) Discuss the dimensions / axis of Porter's Generic Strategy framework and place...
-
Consider the function f and its graph. a. Estimate the zeros of the area function b. Estimate the points (if any) at which A has a local maximum or minimum. c. Sketch a graph of A, for 0 x 10,...
-
The reaction of an organic acid with an alcohol, in organic solvent, to produce an ester and water is commonly done in the pharmaceutical industry. This reaction is catalyzed by strong acid (usually...
-
(a) Describe the primary, secondary, and tertiary structures of proteins. (b) Quaternary structures of proteins arise if two or more smaller polypeptides or proteins associate with each other to make...
-
Both covalent-network solids and ionic solids can have melting points well in excess of room temperature, and both can be poor conductors of electricity in their pure form. However, in other ways...
-
Debate: Survey feedback can be a problematic OD technique because it permits people who are affected by organizational policies to generate data that speak against those policies.
-
Suppose a job enrichment effort in one plant of a manufacturing firm is judged to be very successful. You are the corporate change agent responsible for the project, and you wish to diffuse it to...
-
Describe an example of resistance to change that you have observed. Why did it occur?

Study smarter with the SolutionInn App



