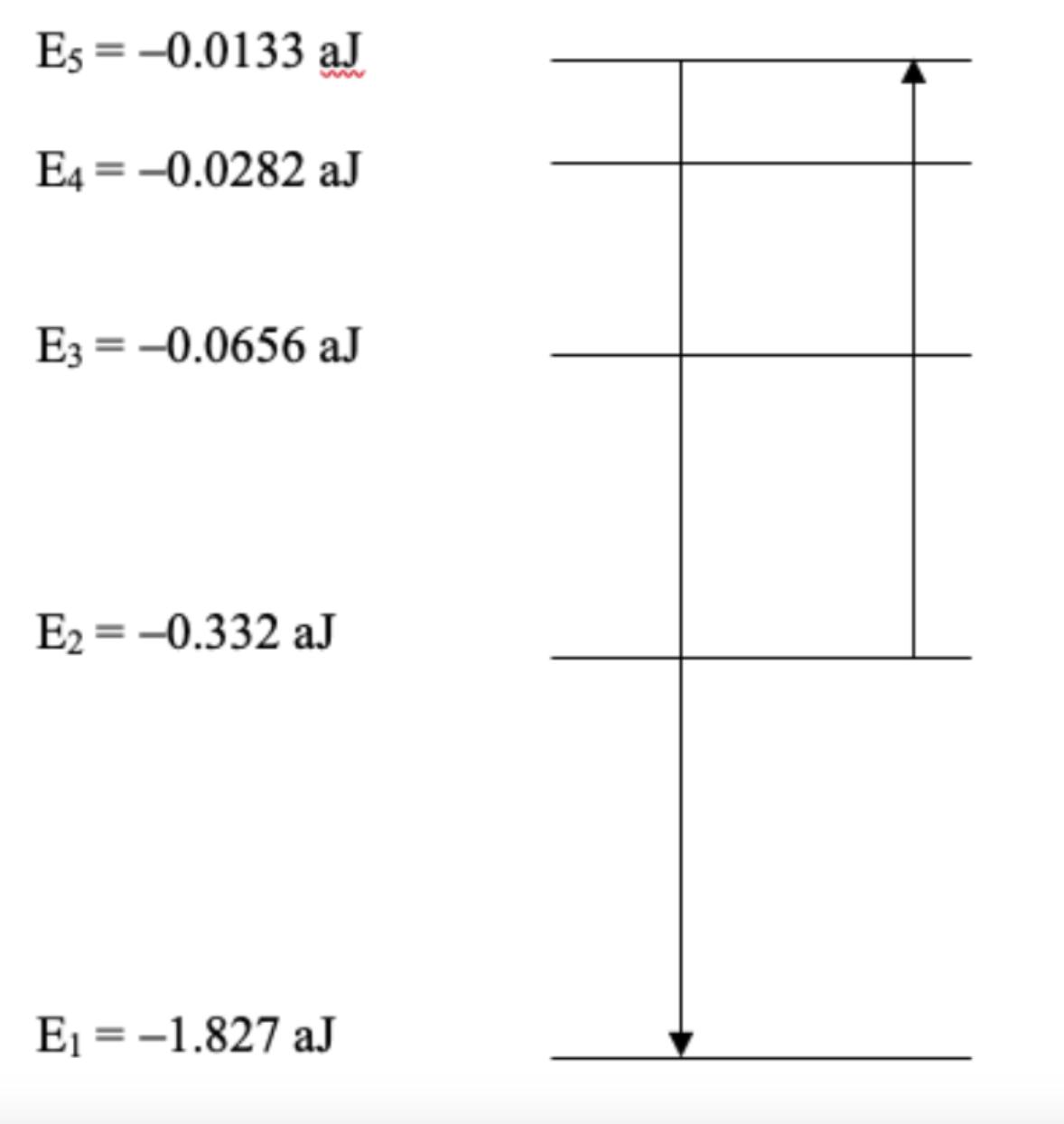
For the atom whose energy levels are depicted below, 653 nm light can be absorbed. Describe the
Fantastic news! We've Found the answer you've been seeking!
Question:
For the atom whose energy levels are depicted below, 653 nm light can be absorbed. Describe the energy level transition experienced by an electron when this happens.
What is the initial energy level and the the final energy level? What is the equation being used and the steps?
Related Book For 

University Physics with Modern Physics
ISBN: 978-0133977981
14th edition
Authors: Hugh D. Young, Roger A. Freedman
Posted Date:




