For the following oxidation and reduction half-reactions, enter the numbers/symbols in the empty boxes to make...
Fantastic news! We've Found the answer you've been seeking!
Question:
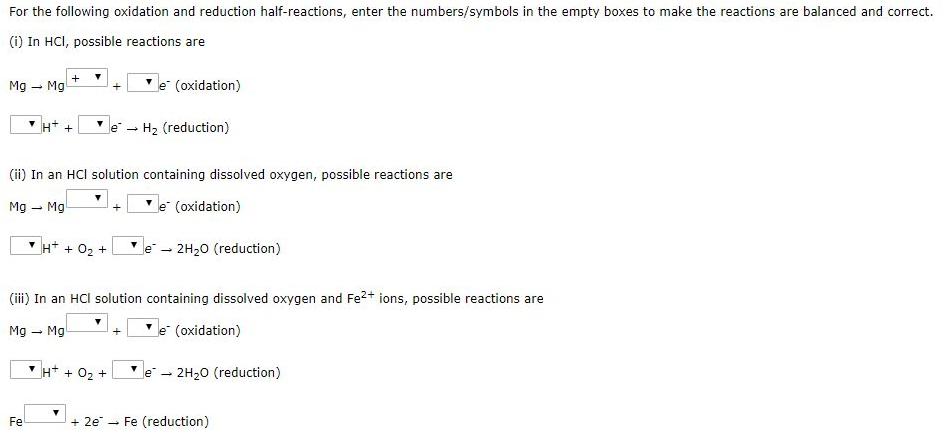
Transcribed Image Text:
For the following oxidation and reduction half-reactions, enter the numbers/symbols in the empty boxes to make the reactions are balanced and correct. (i) In HCI, possible reactions are Mg - Mg e (oxidation) H* + le H2 (reduction) (ii) In an HCI solution containing dissolved oxygen, possible reactions are Mg - Mg e (oxidation) H* + 02 + 2H20 (reduction) le - (iii) In an HCI solution containing dissolved oxygen and Fe?+ ions, possible reactions are Mg – Mg e (oxidation) H+ + 02 + 2H20 (reduction) le - Fe + 2e - Fe (reduction) For the following oxidation and reduction half-reactions, enter the numbers/symbols in the empty boxes to make the reactions are balanced and correct. (i) In HCI, possible reactions are Mg - Mg e (oxidation) H* + le H2 (reduction) (ii) In an HCI solution containing dissolved oxygen, possible reactions are Mg - Mg e (oxidation) H* + 02 + 2H20 (reduction) le - (iii) In an HCI solution containing dissolved oxygen and Fe?+ ions, possible reactions are Mg – Mg e (oxidation) H+ + 02 + 2H20 (reduction) le - Fe + 2e - Fe (reduction)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
In Experiment 1, 200.0 mL of solution containing 10.0 mg of SO42- (from Na2SO4) were treated with excess BaCl2 solution to precipitate BaSO4 containing some coprecipitated Cl_. To find out how much...
-
The observations in the table are dissolved oxygen contents in water. The higher the dissolved oxygen content, the greater the ability of a river, lake, or stream to support aquatic life. In this...
-
The level of dissolved oxygen in water was measured every 2 hours in a river where industrial plants discharge processed waste. Each observation consists of four samples, from which the sample mean...
-
What are some Marketing Strategies for Delivering Objectives Polestar (Car company) has used or using?
-
1. Who prevails and why? 2. Does the corporate veil protect directors in these circumstances?
-
Crossway Apartments, Inc.s unadjusted and adjusted trial balances at April 30, 2012, follow: Requirements 1. Make the adjusting entries that account for the differences between the two trial...
-
The Biltmore National Bank raised capital through the sale of \(\$ 150\) million face value of eight percent coupon rate, ten-year bonds. The bonds paid interest semiannually and were sold at a time...
-
Several items are omitted from the income statement and cost of goods manufactured statement data for two different companies for the month of May: Instructions 1. For both companies, determine the...
-
Actual data (in $1,000) for last five months are provided below. What is the forecast for February, using a four-period weighted moving average with weights of 0.1, 0.2, 0.3 and 0.4 (greater weight...
-
There is a firm in Walla Walla, an isolated site with a labor force of 20 workers. During boom the firm faces a demand curve of P=100-Q. During recession it faces a curve of 80-2Q. Boom and recession...
-
Within the framework of traffic management, which segment pertains to the strategic decision-making process regarding the installation of a new traffic signal at an intersection with the aim of...
-
In what way do legal systems have an impact on global business?
-
use the below info , experience and education to write a cover letter for the following job application: RESPONSIBILITIES 6 0 - 7 0 % - Operates commercial laundry...
-
Analyze and illustrate graphically (Draw graph ) the impact of the following events on the market for young palm tree plants at a nursery. Determine the effect on equilibrium price and quantity. In...
-
Take a case study of any industrial work from Europe made international brand and find the data related to electronic commerce. explain the term according to that industrial impact and please discuss...
-
Within each of the 4 basic, SCM measurement categories (i.e. customer service, internal efficiency, demand flexibility, and product development), there are numerous and varied metrics for...
-
Determine and record pension expense and gains and losses; funding and retiree benefits [LO17-6, 17-7] Actuary and trustee reports indicate the following changes in the PBO and plan assets of Mahomes...
-
Refer to the data for problem 13-36 regarding Long Beach Pharmaceutical Company. Required: Compute each division's residual income for the year under each of the following assumptions about the...
-
A student is given a crucible and asked to prove whether it is made of pure platinum. She first weighs the crucible in air and then weighs it suspended in water (density = 0.9986 g/mL). The readings...
-
Predict the value of (H f (greater than, less than, or equal to zero) for these elements at 25C (a) Br2(g); Br2 (l), (b) I2(g); I2(s)?
-
The solubility of N2 in blood at 37C and at a partial pressure of 0.80 atm is 5.6 3 1024 mol/L. A deepsea diver breathes compressed air with the partial pressure of N2 equal to 4.0 atm. Assume that...
-
Professor Slim Pickens makes the following statement in his Intermediate II Accounting class: The standards of fieldwork provide the foundation to ensure that the financial statements present fairly...
-
Why do you think mandatory job rotation and vacations are effective controls to reduce instances of fraud? In its 2008 Report to the Nation on Occupational Fraud and Abuse, the Association of...
-
Does it surprise you that the audit of ICFR and external audit were ranked at the bottom as effective tools to prevent/detect fraud? Why or why not? In its 2008 Report to the Nation on Occupational...

Study smarter with the SolutionInn App



