For the following reaction, show the product of one mole of reagent adding across the triple...
Fantastic news! We've Found the answer you've been seeking!
Question:
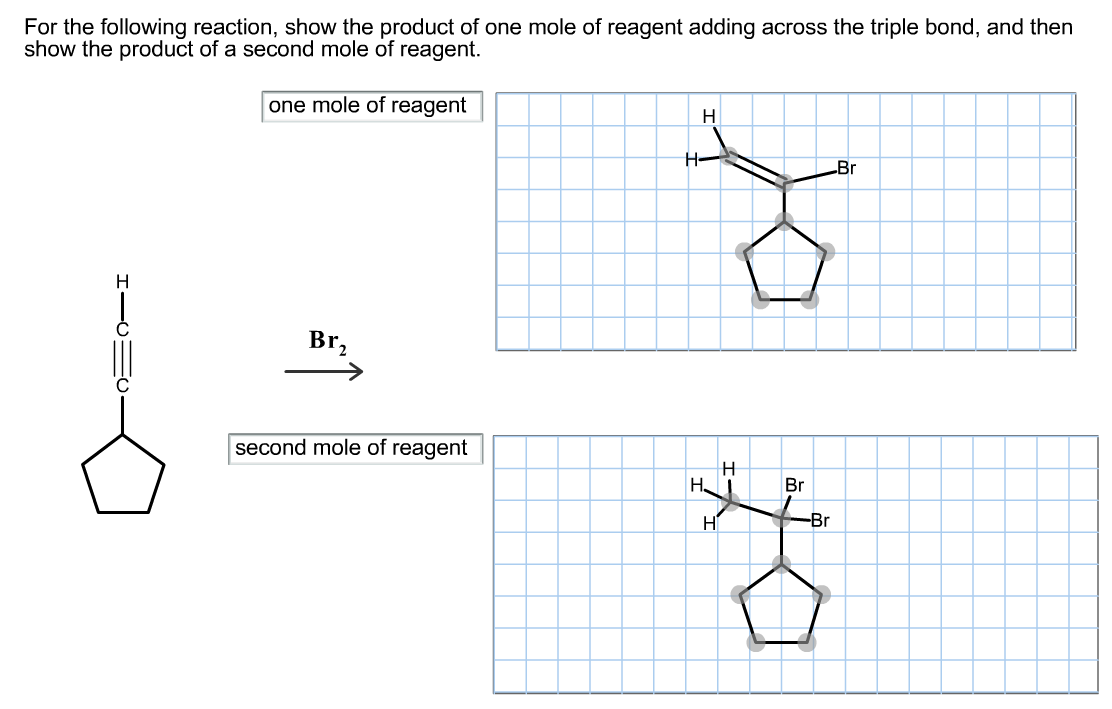
Transcribed Image Text:
For the following reaction, show the product of one mole of reagent adding across the triple bond, and then show the product of a second mole of reagent. one mole of reagent H Br, second mole of reagent H Br X H- H HI H Br -Br For the following reaction, show the product of one mole of reagent adding across the triple bond, and then show the product of a second mole of reagent. one mole of reagent H Br, second mole of reagent H Br X H- H HI H Br -Br
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The equilibrium constant for the following reaction is 1.0 Ã 10-3: Cr'. (aq) + H2EDTA2-(aq)--CrEDT A-(aq) + 2H' (aq) CH2-CO2 02GH CH EDTA N-CH,--CH2- O2C-CH2 CHy-CO2-...
-
The equilibrium constant Kc equals 0.0952 for the following reaction at 227oC. What is the value of Kp at this temperature? CH3OH(g) CO(g) + 2H2(g)
-
The value of Kc for the following reaction at 900oC is 0.28. What is Kp at this temperature? CS2(g) +4H2(g)CH4(g) + 2H2S(g)
-
Harry Bhel carries a business as a sole proprietorship. During its 2022 fiscal period, its first year of operations, the business had cash sales of $123,000. It also has sales on account of $46,000,...
-
The different forms of export financing distribute risks differently between the exporter and the importer. Analyze the distribution of risk in the following export-financing instruments: a....
-
Do you agree that managers and firms really pursue their interests? Why or why not?
-
The price of a non-dividend paying stock is \($19\) and the price of a three-month European call option on the stock with a strike price of \($20\) is \($1.\) The risk-free rate is 4% per annum. What...
-
Bonds 1. Municipal Bonds - Municipal bonds are haircut per Exhibit 1 based on both their time to maturity and scheduled maturity at date of issue. 2. Corporate Bonds - Corporate bonds are haircut...
-
Assistant please, 1) Discuss two impacts to the Database management process by the Data definition language (DDL) and Data manipulation language (DML). 2) Share with a business partner two...
-
Company A is a global company based in the United States that operates in the financial industry. Company A serves its customers with financial products, such as checking accounts, bank cards, and...
-
Use Johnson& Johnson company for this discussion. Analyze selected aspects of the cost structure and key resource elements of the Business Model Canvas to assess the financial performance of that...
-
It is often said that criminal justice employees are unmotivated. Comment on this assertion and discuss possible ways in which the motivational levels of criminal justice workers can be raised. In...
-
5) What is the hexadecimal equivalent for the binary number 1011,0101,0101,1011 6) What is the binary equivalent for the decimal number 391 7) What is the binary equivalent for the hexadecimal number...
-
Your firm is considering an investment in a new one-year project. The project requires an initial investment of $10,000. Based on the probabilities below, the project is expected to produce the...
-
What organizational behavior factors affect criminal justice organizations more than other organizations? What driving forces do you think have instigated the most change in criminal justice...
-
A car starts from rest. It accelerates at 5.4 m/s for 6.8s, coasts for 2.2 s, and then slows down at a rate of 3.8 m/s till the car stops. How far did the car travel from when it started to where it...
-
Our Indiscrete Mathematics course has 28 students from the the College of Arts, 18 of whom are female; 21 students from the the College of Engineering and Informatics, 10 of whom are female; 32...
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
Draw resonance contributors for the following ions: a. b. c. d.
-
The observed rotation of 2.0 g of a compound in 50 mL of solution in a polarimeter tube 50-cm long is +13.4o What is the specific rotation of the compound?
-
Under which of the following reaction conditions would (R)-2-chlorobutane form the most (R)-2-butanol: HO- in 50% water and 50% ethanol or HO- in 100% ethanol?
-
Martinez Company incurred the following costs during 2015 in connection with its research and development activities. Instructions Compute the amount to be reported as research and development...
-
For each of the following studies, identify the type of graph (histogram, time series graph, or scatter diagram) that would be the most appropriate. (You can use more than one graph of each type, for...
-
Explain the difference between artistic-related intangible assets and contract-related intangible assets.

Study smarter with the SolutionInn App



