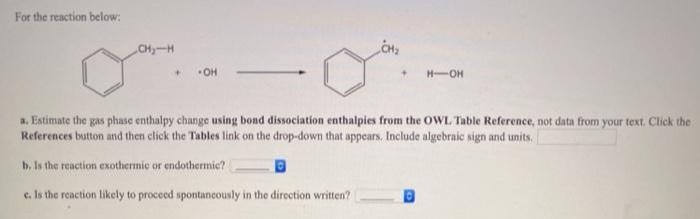
For the reaction below: CHy-H OH H-OH a. Estimate the gas phase enthalpy change using bond...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For the reaction below: CHy-H OH H-OH a. Estimate the gas phase enthalpy change using bond dissociation enthalpies from the OWL Table Reference, not data from your text. Click the References button and then click the Tables link on the drop-down that appears. Include algebraic sign and units. b. Is the reaction exothermic or endothermic? e. Is the reaction likely to proceed spontancously in the direction written? For the reaction below: CHy-H OH H-OH a. Estimate the gas phase enthalpy change using bond dissociation enthalpies from the OWL Table Reference, not data from your text. Click the References button and then click the Tables link on the drop-down that appears. Include algebraic sign and units. b. Is the reaction exothermic or endothermic? e. Is the reaction likely to proceed spontancously in the direction written?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For the reaction below at a certain temperature, it is found that the equilibrium concentrations in a 5.00-L rigid container are [H2] = 0.0500 M, [F2] = 0.0100 M, and [HF] = 0.400 M. H2(g) + F2(g) ...
-
The energy profile diagram represents (a) An endothermic reaction (b) An exothermic reaction (c) A fast reaction (d) A termolecular reaction A + B Progress of reaction >
-
1. Is the Haber process an exothermic or endothermic reaction? (2 pts) 2. Write a balanced thermochemical equation with phase labels for the Haber process with the heat energy as part of the...
-
You are an audit supervisor of PricewaterhouseCoopers (PwC) and are planning the audit of your client, Blister Pharmaceuticals co. which manufactures and imports sanitary and cleaning products...
-
In the lingo of craps-shooters (where two dice are tossed and the underlying sample space is the matrix pictured in Figure 2.2.1) is the phrase making a hard eight. What might that mean?
-
Ferraro, Inc. established a share-appreciation rights (SAR) program on January 1, 2010, which entitles executives to receive cash at the date of exercise for the difference between the market price...
-
Many researchers are interested in the transcription of protein-encoding genes in eukaryotes. Such researchers want to study mRNA. One method that is used to isolate mRNA is column chromatography....
-
Pauline Found Manufacturing, Inc., is moving to kanbans to support its telephone switching- board assembly lines. Determine the size of the kanban for subassemblies and the number of kanbans needed....
-
Q6). For the Given Functions determine the following a). f(x) = 3 x2+2x-1 What is f(-2). Show your work 0 What is a). f(-2)= b). f(0), c). f(-1)
-
A contestant on the hit reality television show Top Bartender was asked to mix a variety of drinks, each consisting of 4 fluid ounces. No other ingredients were permitted. She was given the following...
-
Estimate the approximate age of the company's long-term assets (show your calculations). Constellation Brands (amounts in millions) Property and Equipment: 9257.10 in 2023, 8011.90 in 2022, 7555.80...
-
While effective resourcing and talent management strategies can drive business success, discuss the key challenges that telecommunication company may face?
-
Which one of the following situations correctly describes how to account for a change in estimate? A. A client amortizes computer hardware over four years and computer software over two years. While...
-
What are the skills needed to be a successful project manager.
-
Dr. Leroy is budgeting for the upcoming year. As he meets with the nursing executives, he explains how every position needs to be justified, each expenditure line item analyzed to determine necessity...
-
A great project manager, must be a strategic business partner fully vested in organizational success and be able to roll with inevitable setbacks. There are a number of demands that are unique to...
-
Assessment 2.0: Construction historical context For a line or route for which you are familiar, undertake research, and compile a report (min 500 words) to identify the historical context of the...
-
On average there are four traffic accidents in a city during one hour of rush-hour traffic. Use the Poisson distribution to calculate the probability that in one such hour there arc (a) No accidents...
-
The isoelectric point of an amino acid is the pH at which the molecule has no net charge. For glycine, that would be the pH at which virtually all glycine molecules are in the form +H3NCH2CO2-. If we...
-
Describe, in general, the structures of ionic solids. Com-pare and contrast the structures of sodium chloride and zinc sulfide. How many tetrahedral holes and octahedral holes are there per closest...
-
Consider the following statements. Write out an example reaction and K expression that are associated with each statement. a. The autoionization of water. b. An acid reacts with water to produce the...
-
Using the knowledge you gained from your interview in mini case 1 , design an interface for the individual you interviewed. What interface design modifications are you making so that the program will...
-
The capital market line, CML, is the graph of the risk and return of portfolio combinations consisting of the risk-free asset and: A. Any risky portfolio. B. The market portfolio. C. The leveraged...
-
Relative to portfolios on the CML, any portfolio that plots above the CML is considered: A. Inferior. B. Inefficient. C. Unachievable.

Study smarter with the SolutionInn App


