For the reaction below, the thermodynamic equilibrium constant is K= 1.3310- at 45 C. NH4CONH(s) ...
Fantastic news! We've Found the answer you've been seeking!
Question:
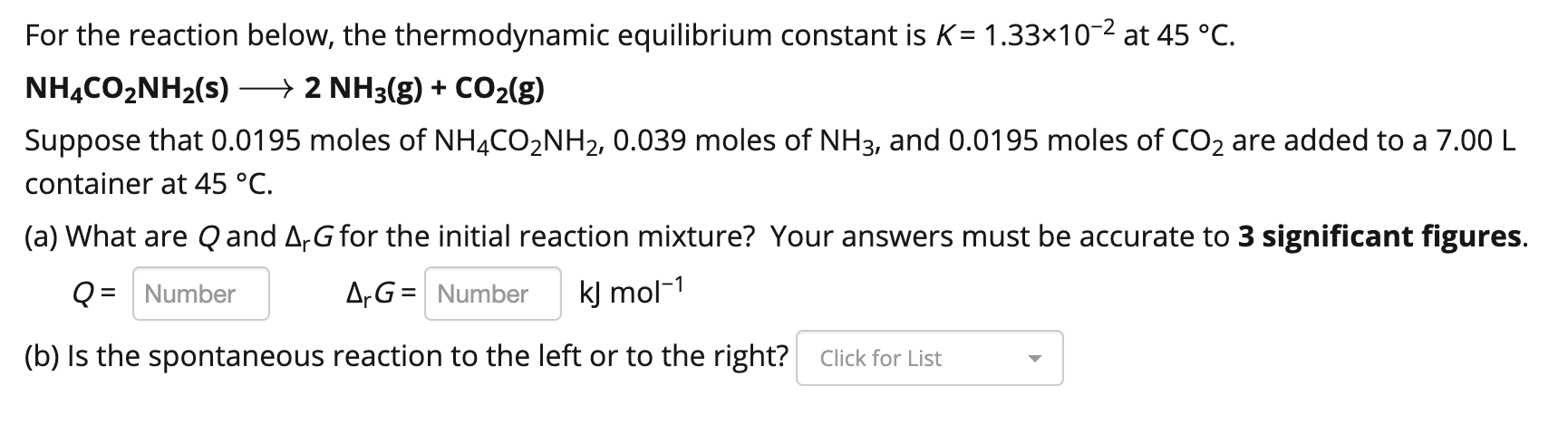
Transcribed Image Text:
For the reaction below, the thermodynamic equilibrium constant is K= 1.33×10-² at 45 °C. NH4CO₂NH₂(s) → 2 NH3(g) + CO₂(g) Suppose that 0.0195 moles of NH4CO₂NH₂, 0.039 moles of NH3, and 0.0195 moles of CO2 are added to a 7.00 L container at 45 °C. (a) What are Q and ArG for the initial reaction mixture? Your answers must be accurate to 3 significant figures. Q = Number ArG = Number kJ mol-1 (b) Is the spontaneous reaction to the left or to the right? Click for List For the reaction below, the thermodynamic equilibrium constant is K= 1.33×10-² at 45 °C. NH4CO₂NH₂(s) → 2 NH3(g) + CO₂(g) Suppose that 0.0195 moles of NH4CO₂NH₂, 0.039 moles of NH3, and 0.0195 moles of CO2 are added to a 7.00 L container at 45 °C. (a) What are Q and ArG for the initial reaction mixture? Your answers must be accurate to 3 significant figures. Q = Number ArG = Number kJ mol-1 (b) Is the spontaneous reaction to the left or to the right? Click for List
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Suppose 72.3 J of heat are added to a 101-g piece of aluminum at 20.5 C. What is the final temperature of the aluminum?
-
For the reaction below at a certain temperature, it is found that the equilibrium concentrations in a 5.00-L rigid container are [H2] = 0.0500 M, [F2] = 0.0100 M, and [HF] = 0.400 M. H2(g) + F2(g) ...
-
If 2200 J of heat are added to a 190-g object, its temperature increases by 12 C. (a) What is the heat capacity of this object? (b) What is the object's specific heat?
-
Consider a stylized two-period model with banking. The aggregate abatement cost function in period t is given by C(E)= (a t be) 2 /2b with a 1 < a 2 is D(E)=dE 2 /2. (a) Determine the optimal...
-
The introduction to this experiment shows the general equation for an etherification reaction. Use the equation as a guide to write down balanced equations representing each of the esterification...
-
If the house is currently worth $355,000 and most lenders are willing to lend up to 90% of home value, how much excess equity can the Sommers cash out?
-
Continuing to focus on evidence associated with the act, concealment, and conversion, use the evidentiary material to continue the examination. In addition, the examiner also starts to think of terms...
-
Assume that you have an after-tax cost of capital of 10 percent. Compute the net present value of each of the five projects listed in the following table. Rank the above projects from 1 (best for...
-
A manager could increase his or her accounting income-based bonus by Multiple choice question. delaying discretionary expenses. delaying shipments of its products. disclosing additional contingent...
-
"Part 1: The Performance Lawn Equipment database contains data needed to develop a pro forma income statement. Dealers selling PLE products all receive 18% of sales revenue for their part of doing...
-
If a recipe calls for 600 grams of chocolate how many ounces of chocolate do you need to use? You have just converted a recipe for a root vegetable soup. The recipe is now calling for 6 cups of heavy...
-
Suppose you are the money manager of a $4.07 million investment fund. The fund consists of four stocks with the following investments and betas: Stock Investment Beta A $240,000 1.50 B 560,000 (0.50)...
-
What do you think the three biggest challenges are that companies face when managing their inventory mix and quantities? Your Opinion Voice your opinion in the main post and state three reasons to...
-
Find the function value, if possible. (If an answer is undefined, enter UNDEFINED.) f(x) = (4x+3, x < 0 = 4x+4, x 0 (a) f(-1) (b) f(0)
-
A $1,000 face value bond is currently selling for $800 (the current Price is $800). The required rate is 6% and it has 20 years to maturity. It pays an annual coupon. What is the coupon payment in...
-
Two blocks of masses m = 1.96 kg and m2 = 5.14 kg are each released from rest at a height of h = 4.94 m on a frictionless track, as shown in the figure, and undergo an elastic head-on collision. m m2...
-
First, identify a service industry of mine is, General Motors. Then, in a short paper, do the following: Why does General Motors start production on the Camaro and, after a few years, decide to stop...
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
Consider a gaseous binary compound with a molar mass of 62.09 g/mol. When 1.39 g of this compound is completely burned in excess oxygen, 1.21 g of water is formed. Determine the formula of the...
-
For the reaction 2A + B products a friend proposes the following mechanism: A + B M A + M products a. Assuming that the second step is the rate-determining step and the first step is a fast...
-
Carbonate buffers are important in regulating the pH of blood at 7.40. If the carbonic acid concentration in a sample of blood is 0.0012 M, determine the bicarbonate ion concentration required to...
-
The codon change (Gly-12 to Val-12) in human H-ras that converts it to oncogenic H-ras has been associated with many types of cancers. For this reason, researchers would like to develop drugs to...
-
Outline the general strategy used in metagenomics.
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A muscle-specific gene was cloned and then subjected to promoter bashing. As shown...

Study smarter with the SolutionInn App


