For the reaction; CH3COOH + HO CH3COO + H3O+ The concentration of the products and...
Fantastic news! We've Found the answer you've been seeking!
Question:
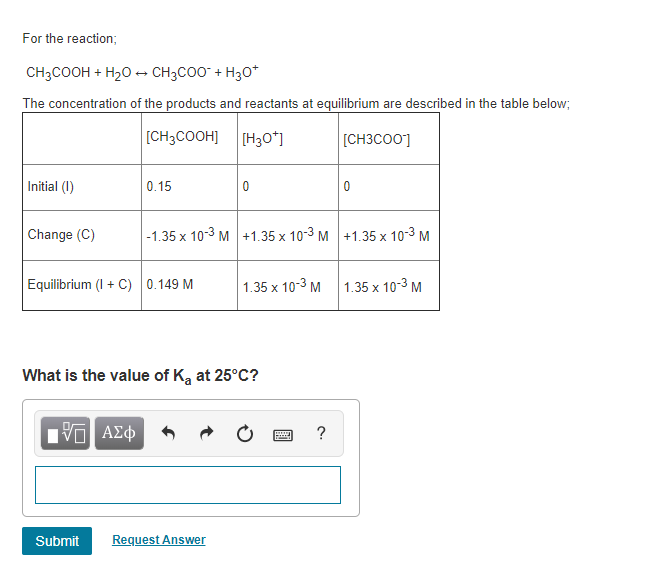
Transcribed Image Text:
For the reaction; CH3COOH + H₂O → CH3COO + H3O+ The concentration of the products and reactants at equilibrium are described in the table below; [CH3COOH] [H3O+] Initial (1) Change (C) 0.15 Equilibrium (I + C) 0.149 M ΙΠ ΑΣΦ Submit What is the value of K₂ at 25°C? -1.35 x 10-3 M +1.35 x 10-3 M +1.35 x 10-³ M 1.35 x 10-3 M Request Answer wwwww [CH3C00] ? 0 1.35 x 10-3 M For the reaction; CH3COOH + H₂O → CH3COO + H3O+ The concentration of the products and reactants at equilibrium are described in the table below; [CH3COOH] [H3O+] Initial (1) Change (C) 0.15 Equilibrium (I + C) 0.149 M ΙΠ ΑΣΦ Submit What is the value of K₂ at 25°C? -1.35 x 10-3 M +1.35 x 10-3 M +1.35 x 10-³ M 1.35 x 10-3 M Request Answer wwwww [CH3C00] ? 0 1.35 x 10-3 M
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the value of K if the concentrations at equilibrium are shown for the reaction below? [D] = 4.4 x 10 -5 M [C] = 2.2 x 10 -2 M [B] = 9.4 x 10 -3 [A] = 1.2 x 10 -2 D(ag) + 2C(ag) = 2B(aq) +...
-
At 700 K the equilibrium constant for the reaction Is Kp= 0.76. A flask is charged with 2.00 atm of CCl4, which then reaches equilibrium at 700 K. (a) What fraction of the CCl4 is converted into C...
-
The equilibrium reaction as: CH4 C + 2H2, has ln K = -0.3362 at 800 K and ink = -4.607 at 600 K. By noting the relation of K to temperature shows how you would interpolate ln K in...
-
Question 2: Consider the market for Florida oranges. The demand for Florida oranges is given by the inverse demand function p = 70-2Q The market cost function for firms that sell Florida oranges is...
-
Go to this texts Web site at academic. cengage.com/blaw/clarkson and select Chapter 51. Click on Video Questions and view the video titled Accountants Liability. Then answer the following questions....
-
In general, what are the principles on which the Modified Accelerated Cost Recovery System (MACRS) is based?
-
\(1,000 \div 5^{3}-7 \times 8^{4}\) Perform the indicated calculation.
-
Tax Agency Fund. The county collector of Lincoln County is responsible for collecting all property taxes levied by funds and governments within the boundaries of the county. To reimburse the county...
-
High Tec Manufacturing uses a job order cost system withperpetual inventory for its specialized jobs. At the beginning of2020, there is one completed job on hand, Job Z29 with a cost of$134,000. Al 1...
-
Kevin has met with you and Brenda. Brenda has explained his current duties as an enduring power of attorney holder. She has also provided Kevin with a copy of Norman's will and briefly explained his...
-
For the last six weeks, we have been exploring the concepts of human behavior relevant to the successful operation of an organization. You've analyzed problems and made recommendations for a variety...
-
If we consider this real interest rate as the main interest rate that the Fed targets, do you think the Fed played a role in causing the recession, or trying to prevent it, or neither? Explain your...
-
Noble cause corruption follows the teleological model of ethics. The approach emphasizes "good ends" and is a product of police officers' "overinvestment" in their work. Police officers believe that...
-
1) Calculate the Operating Cycle. 2) Calculate the Cash Cycle 3) List an idea to improve the Operating Cycle 4) List an idea to improve the Cash Cycle
-
How does interest rate risk impact the balance sheet of financial institutions, and what strategies do banks employ to hedge against interest rate volatility, especially during periods of monetary...
-
Find the determinant. 34 A= 57
-
The Annotated Bibliography The purpose of this assignment is to demonstrate progress in compiling research and to indicate a methodology for organizing your research sources. Include the following in...
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
Define the terms complex ion, ligand, and coordination number. Use an example to illustrate the use of these terms.
-
Calculate the pressure of water vapor at 120.0oC if 1.000 mol of water vapor occupies 32.50 L. Use the van der Waals equation (see Table 5.7 for data). Compare with the result from the ideal gas law.
-
Methyl acetate reacts in acidic solution. The rate law is first order in methyl acetate in acidic solution, and the rate constant at 25oC is 1.26 104/s. How long will it take for 65% of the methyl...
-
Consider the family of differential equations \(x^{\prime}=x^{3}+\delta x^{2}-\mu x\). a. Sketch a bifurcation diagram in the \(x \mu\)-plane for \(\delta=0\). b. Sketch a bifurcation diagram in the...
-
For each problem, determine equilibrium points, bifurcation points, and construct a bifurcation diagram. Discuss the different behaviors in each system. a. \(y^{\prime}=y-\mu y^{2}\) b....
-
Plot phase portraits for the Lienard system \[\begin{aligned} & x^{\prime}=y-\mu\left(x^{3}-x ight) \\ & y^{\prime}=-x \end{aligned}\] for a small and a not so small value of \(\mu\). Describe what...

Study smarter with the SolutionInn App



