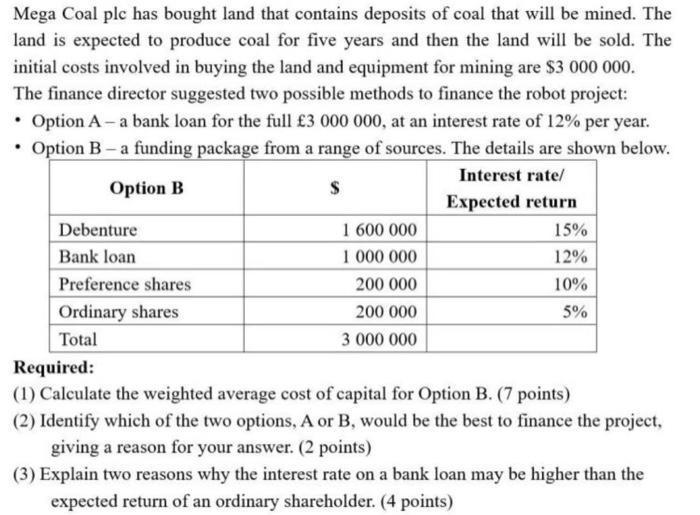
Mega Coal ple has bought land that contains deposits of coal that will be mined. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Mega Coal ple has bought land that contains deposits of coal that will be mined. The land is expected to produce coal for five years and then the land will be sold. The initial costs involved in buying the land and equipment for mining are $3 000 000. The finance director suggested two possible methods to finance the robot project: • Option A - a bank loan for the full £3 000 000, at an interest rate of 12% per year. • Option B-a funding package from a range of sources. The details are shown below. Interest rate/ Option B Expected return Debenture Bank loan Preference shares Ordinary shares Total 1 600 000 1 000 000 200 000 200 000 3 000 000 15% 12% 10% 5% Required: (1) Calculate the weighted average cost of capital for Option B. (7 points) (2) Identify which of the two options, A or B, would be the best to finance the project, giving a reason for your answer. (2 points) (3) Explain two reasons why the interest rate on a bank loan may be higher than the expected return of an ordinary shareholder. (4 points) Mega Coal ple has bought land that contains deposits of coal that will be mined. The land is expected to produce coal for five years and then the land will be sold. The initial costs involved in buying the land and equipment for mining are $3 000 000. The finance director suggested two possible methods to finance the robot project: • Option A - a bank loan for the full £3 000 000, at an interest rate of 12% per year. • Option B-a funding package from a range of sources. The details are shown below. Interest rate/ Option B Expected return Debenture Bank loan Preference shares Ordinary shares Total 1 600 000 1 000 000 200 000 200 000 3 000 000 15% 12% 10% 5% Required: (1) Calculate the weighted average cost of capital for Option B. (7 points) (2) Identify which of the two options, A or B, would be the best to finance the project, giving a reason for your answer. (2 points) (3) Explain two reasons why the interest rate on a bank loan may be higher than the expected return of an ordinary shareholder. (4 points)
Expert Answer:
Answer rating: 100% (QA)
1 To calculate the weighted average cost of capital WACC for Option B we need to first determine the proportion of each type of financing in the overa... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
The Pitts Limited had EBITDA of $900 million in 2019. The depreciation amount for the period $300m. Firms increased investment in fixed assets by $400m from 2018. Firms overall investment in working...
-
You and a partner are asked to complete a lab entitled "Oxides of Ruthenium" that is scheduled to extend over two lab periods. The first lab, which is to be completed by your partner, is devoted to...
-
In this simulation, you are asked to address questions related to the accounting for leases. Prepare responses to all parts. (Round amounts to the nearestcent.) KWW Professional Simulation Time...
-
Find the inverse function f -1 of function f. Find the range of f and the domain and range of f -1 . 2 S(x) = 2 cos(3x + 2): sxs+ 3
-
The diagrams below show the market for gasoline in two countries, Midas and Neptune. In Midas, demand is perfectly inelastic; in Neptune, demand is relatively elastic. In both countries, supply is...
-
The debits and credits from three related transactions are presented in the following creditor's account taken from the accounts payable ledger. Describe each transaction, and identify the source of...
-
An inward flow radial turbine (see Fig. P5.136) involves a nozzle angle, \(\alpha_{1}\), of \(60^{\circ}\) and an inlet rotor tip speed, \(U_{1}\), of \(30 \mathrm{ft} / \mathrm{s}\). The ratio of...
-
Accounts Payable Confirmations. Partners Clark and Kent, both CPAs, are preparing their audit plan for the audit of accounts payable on Marlboro Corporations annual audit. Saturday afternoon they...
-
Mike's total RRSP contribution room for the current year is $8,000, while his wife Maria's room is $2,000. Mike has decided to contribute $5,000 to his own RRSP and $3,000 to a spousal RRSP for...
-
1. Figure 1 reproduces John's notes on Pioneer Gypsum and Global Mining. Calculate the expected return, risk premium, and standard deviation of a portfolio invested partly in the market and partly in...
-
Write a paper on a research topic relating to Data Management.
-
Why the simple model of exchange is a special case of the static Arrow- Hurwicz model?
-
At each moment t and the price of the second good change according to equations: t = [0, 20] a consumer's income, the price of the first good
-
Why are only feasible price trajectories taken into consideration? How are they defined in both versions of the dynamic Arrow-Hurwicz model?
-
a. Did the court violate Aron Goldbergers First Amendment right to freedom of religion by forcing him to pay child support? b. Assume you work for the law firm that represents Esther Goldberger. To...
-
There are given a power utility function (x)n = ax x, a, a, > 0, +
-
What do you believe is the purpose of financial accounting, and who are users of that information? Who is generally credited with "inventing" modern accounting, and when and where did he create it?
-
On October 31 Juanita Ortega, owner of Outback Guide Service, received a bank statement dated October 30. Juanita found the following: 1. The checkbook has a balance of $2,551.34. 2. The bank...
-
What is the boiling point of a 0.10 M solution of NaHSO4 if the solution has a density of 1.002 g > mL?
-
Complete and balance the following equations: (a) Mg3N2(s) + H2O(l) (b) C3H7OH(l) + O2(g) (d) AlP(s) + H2O(l) (e) Na2S(s) + HCl(aq) A
-
For each of the following metals, write the electronic configuration of the atom and its ion: (a) Ru, (b) Mo, (c) Co.Draw the crystal-field energy-level diagram for the d orbitals of an octahedral...
-
The PFD in Figure 11.63 shows a process in which two liquid products, A and B, are produced from a feed stream of raw material R. In the process, the reactor feed is preheated to \(300^{\circ}...
-
Consider a process with the following streams: (a) Compute \(\Delta T_{\text {thres }}\) as well as the minimum external heating and cooling requirements as a function of \(\Delta T_{\text {min }}\)....
-
Design a HEN to meet the MER targets for \(\Delta T_{\text {min }}=10^{\circ} \mathrm{C}\) and \(N_{H X, \text { min }}\) for a process involving five hot streams and one cold stream as introduced by...

Study smarter with the SolutionInn App


