a) What is the rate law for this reaction? b) What is the rate constant for...
Fantastic news! We've Found the answer you've been seeking!
Question:

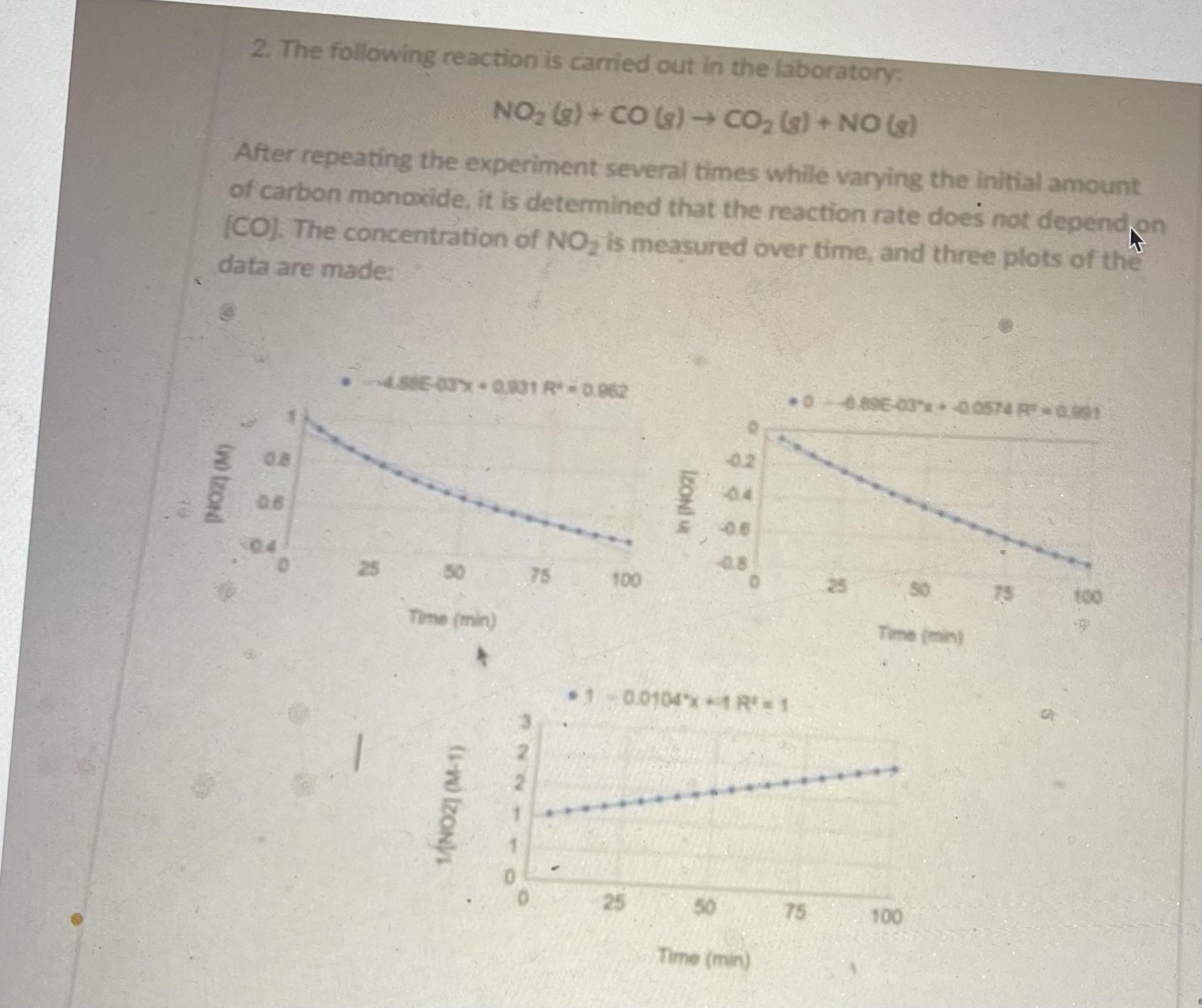
Transcribed Image Text:
a) What is the rate law for this reaction? b) What is the rate constant for this reaction? Don't forget units! )A two-step mechanism is proposed for this reaction. Write the chemical equation for the second step. Step 1:2 NO2 (g) - NO, (3) + NO (3) Step 2: ?? d) Which step has the larger activation barrier, step 1 or step 2? Explain your answer in 1-2 sentences. 2. The following reaction is carried out in the laboratory NO2 G) + CO (s) CO, (s)+ NO (e) After repeating the experiment several times while varying the initial amount of carbon monaxide, it is determined that the reaction rate does not depend on [CO]. The concentration of NO, is measured over time, and three plots of the data are made: 4.88E-03x 0831 R0.962 06896-03* 0.0574 R 0.991 02 0.8 04 08 0.6 04 08 25 50 75 100 25 50 75 100 Time (min) Time (min) 0.0104x1 R= 1 25 50 75 100 Time (min) IZONI M 1ÁNOZI (M-1) ino tzond a) What is the rate law for this reaction? b) What is the rate constant for this reaction? Don't forget units! )A two-step mechanism is proposed for this reaction. Write the chemical equation for the second step. Step 1:2 NO2 (g) - NO, (3) + NO (3) Step 2: ?? d) Which step has the larger activation barrier, step 1 or step 2? Explain your answer in 1-2 sentences. 2. The following reaction is carried out in the laboratory NO2 G) + CO (s) CO, (s)+ NO (e) After repeating the experiment several times while varying the initial amount of carbon monaxide, it is determined that the reaction rate does not depend on [CO]. The concentration of NO, is measured over time, and three plots of the data are made: 4.88E-03x 0831 R0.962 06896-03* 0.0574 R 0.991 02 0.8 04 08 0.6 04 08 25 50 75 100 25 50 75 100 Time (min) Time (min) 0.0104x1 R= 1 25 50 75 100 Time (min) IZONI M 1ÁNOZI (M-1) ino tzond
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Why is the T-bill's return independent of the state of the economy? Do T-bills promise a completely risk-free return? (2) Why are Alta Industries' returns expected to move with the economy whereas...
-
What is the rate of energy radiation per unit area of a blackbody a\ a temperature of? (a) 273 K and (b) 2730 K?
-
The rate law for a reaction can be determined only from experiment and not from the balanced equation. Two experimental procedures were outlined in this chapter. What are these two procedures?...
-
To enable a process to Wait(condition) within the monitor, which one of the following is TRUE? All of mentioned Semaphore must be used Condition variables must be used as boolean objects A condition...
-
Burnett Company produces two types of gears: Model 12 and Model 15. Market conditions limit the number of each gear that can be sold. For Model 12 no more than 15,000 units can be sold, and for Model...
-
A disk drive manufacturer estimates that in five years a storage device with 1 terabyte of capacity will sell with probability 0.5, a storage device with 500 gigabytes capacity will sell with a...
-
Washington Mutual Insurance Company issued an \(\$ 80,000,7 \%, 10\)-vear bond payable at a price of 110 on January 1, 2009. Journalize the following transactions for Washington. Include an...
-
Rembrandt Frame Company prepared the following sales budget for the current year: At the end of December 2008, the following unit sales data were reported for the year: For the year ending December...
-
3. Ch4. Cash Discount/Dating Terms (2.5pts). Using the information given in the table, find cash discount due date. ****Note: We are using 30 day month method where all months are considered to have...
-
You have just been hired as a financial analyst for Lydex Company, a manufacturer of safety helmets. Your boss has asked you to perform a comprehensive analysis of the company's financial statements,...
-
You own a portfolio that isinvested 50 percent in stock A, 15 percent in stock B, and theremainder in stock C. The expected returns on these stocks are14.45 percent, 15.6 percent, and 12.33 percent,...
-
Find the input impedance Z 1 = V 1 /I 1 in the coupled circuit of Fig. 14-36. For s = j. 1 M = H Fig. 14-36
-
What is the difference between pound-mass and pound-force?
-
A population has mean = 53 and standard deviation = 34. Find the value that has 35% of the population above it.
-
What is a steady-flow process?
-
Is it correct to say that an object with a smooth surface produces very low form drag?
-
What effect, if any, would the proposed change have on Vance's income tax expense, net income, and cash flows? A FIFO 1500000 $ LIFO 80000 Income tax expense Net income 600000 +A +A $ 320000
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
Ammonia burns in the presence of a copper catalyst to form nitrogen gas. 4NH3(g) + 3O2(g) 2N2(g) + 6H2O(g); H = 1267 kJ What is the enthalpy change to burn 35.8 g of ammonia?
-
When ammonia is first added to a solution of copper(II) nitrate, a pale blue precipitate of copper(II) hydroxide forms. As more ammonia is added, however, this precipitate dissolves. Describe what is...
-
The [Fe(H2O)6]3+ ion has a pale purple color, and the [Fe(CN)6]3 ion has a ruby-red color. What are the approximate wavelengths of the maximum absorption for each ion? Is the shift of wavelength in...
-
Execute an ANOVA to compare the attitudes, opinions, purchase and recommend likelihood of groups based on level of education completed and number of music products owned. What are the interesting...
-
Execute an ANOVA to compare the likelihood to purchase and recommend vinyl products for groups based on gender and age. What are the interesting findings?
-
Are there other topics that should be added to this questionnaire?

Study smarter with the SolutionInn App


