Be sure your stirring rod is washed and dried. Stir well. This is your starting citric acid
Question:

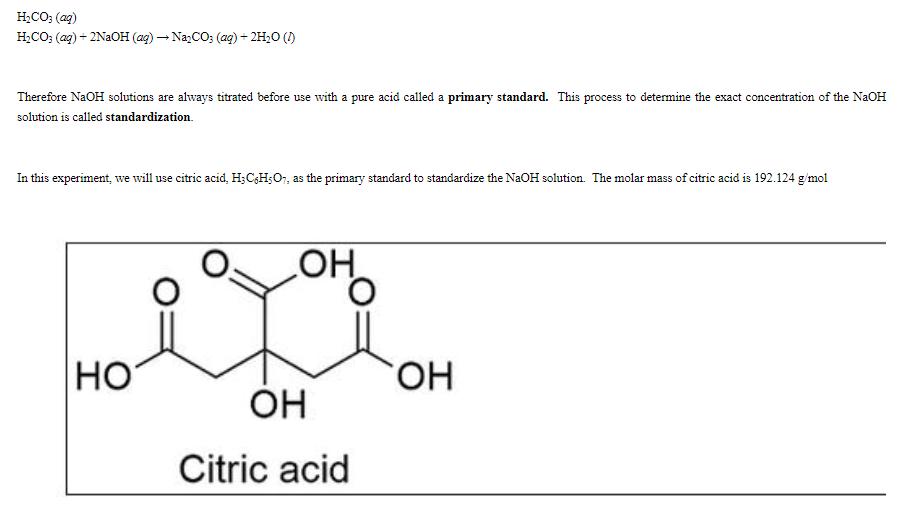

- Be sure your stirring rod is washed and dried. Stir well.
- This is your starting citric acid solution. You will need to calculate the molarity of this citric acid solution and use that to standardize the NaOH.
Note: Be careful with the measurements of citric acid and the water. Based on the mass of citric acid and the volume of solution, you will calculate the exact molarity of the citric acid solution. This exact molarity of the citric acid solution is needed to determine the molarity of the NaOH solution.
- Once the citric acid is dissolved and the solution mixed thoroughly, measure out approximately 20.0 mL of the citric acid solution and place it in the small empty100-mL beaker.
- Record the exact volume on the lab report under Trial 1 column.
- Add 2 drops of phenolphthalein to the citric acid solution in the 100-mL beaker.
- Swirl to mix.
- Fill a 10-mL graduated cylinder exactly to the 10.0 mL mark with your NaOH solution. Record the initial volume of the NaOH solution on your lab report. Always be sure to record the correct number of decimal places which in this case would be one decimal.
- Titrate by using a clean plastic pipette dropper to transfer the NaOH solution from the graduated cylinder dropwise to the citric acid solution in the 100-mL beaker while swirling.
Lab technique tip: Gently swirl the 100-mL beaker while the NaOH solution drips from the tip of the pipette dropper. Be careful not to splash out the solution.
- Stop the titration when the mixture changes from colorless to faintly pink (this is the endpoint.) The titration is finished when the faint pink color persists for at least 30 seconds. See the picture below.
Lab Technique Tip: Do not over titrate. Add just enough NaOH to get a faint pink color. As you add the NaOH you will see the pink color appear, but it will not persist after swirling. As you near the endpoint, the pink color takes longer to disappear until one drop turns the entire solution pink. The middle picture below shows what the solution should look like when the titration is finished. If you add too much NaOH, you will get a dark pink color. You want the faintest pink perceptible.
Before the endpoint Endpoint Beyond the endpoint
- Once you reach the endpoint and stop the titration, return any unused NaOH in the pipette to the 10-mL graduated cylinder.
- Read the volume as accurately as possible. Record the final volume of the NaOH solution in the 10-mL graduated cylinder to the proper number of digits.
Note: The sodium hydroxide used in the trial will be the difference between the initial volume of NaOH in the graduated cylinder and the final volume of the NaOH in the graduated cylinder.
- Dispose of the pink titration solution in the sink with water and rinse the beaker with distilled water.
- Perform two more trials with the citric acid by repeating Steps 8-17.
- Calculate the NaOH concentration from each trial.
- Calculate the average NaOH concentration of all three trials.
Note: This NaOH solution is now your standardized NaOH solution. The concentration of this solution is the average concentration of all three trials. This is necessary information in order to determine the unknown in the next part. Do not waste the NaOH solution, as you will not be able to mix up additional NaOH if you run out of the standardized solution.
Part II: Determination of % Citric Acid in Unknown Impure Sample
- Obtain the solid Unknown.
- Rinse out the remaining citric acid solution in the 100-mL beaker from Part I. Clean and dry the 100-mL beaker
- Place the clean and dry plastic weigh boat on your balance and tare it.
- Weigh out exactly 0.900 g of your unknown and record the mass to the proper number of digits.
- Transfer the unknown into the clean and dry 250-mL beaker.
- Measuring the volume very carefully with the graduated cylinder, add 100.0 mL of distilled water to the 250-mL beaker and stir well. This is your unknown impure citric acid solution.
- Carry out three titration trials on the unknown using the same techniques that you used in Part I by repeating steps 8-17 of Part I.
- Calculate the % citric acid in your unknown sample for each trial.
- Calculate the average % citric acid in your unknown sample.
Calculations:
Part I Standardization of NaOH using Anhydrous Citric Acid
First, we need the starting molarity of the citric acid solution we mixed. A student used 0.16 g of citric acid and 100.0 mL of water to make 100.0 mL of solution.
starting moles of citric acid = mass of citric acid x 1 mole citric acid192.124 g citric acid
= 0.16 g citric acid x 1 mole citric acid192.124 g citric acid
= 8.328 x 10−4 moles citric acid
molarity of citric acid solution = mole citric acid0.1000 L of solution
= 8.328 x 10-4 moles citric acid 0.1000 L of solution
= 8.328 x 10−3 M citric acid
Once we have the molarity of citric acid solution, we will use the volume of citric acid solution in each trial to determine the moles of citric acid in each titration trial. This is our starting point in solution stoichiometry. The student used 20.0 mL of the citric acid solution in the trial. (Always watch the units.)
mole of citric acid in each trial = Molaritycitric acid x Lcitric acid
= (8.328 x 10−3 M)(0.0200L)
= 1.6656 x 10−4 mole of citric acid
According to neutralization reaction of citric acid with NaOH, one mole of citric acid reacts with three mole of NaOH. This is our mole-to-mole ratio. Therefore, at the equivalence point:
1 mole citric acid = 3 moles NaOH
mole NaOH in each trial = moles of citric acid x 3 mole NaOH1 mole citric acid
Since we know the both the moles and volume of NaOH used in the trial, we can use the formula for molarity to determine the molarity of the standardized NaOH solution of each trial:
molarity of NaOH in each trial = mol NaOH L of NaOH solution
Calculation Tip: Always pay attention to units in these calculations.
Once we have calculated the molarity of NaOH for each trial, we can calculate the average M of the NaOH solution.
average M of NaOH = Trial 1 + Trial 2 + Trial 33
PART II: Determination of % Citric Acid in Unknown Impure Sample
Here we begin with the average molarity of our standardized NaOH solutions. From the average molarity, we can determine the moles of NaOH used in the titration trial of the unknown.
moles of NaOH used in each trial = MNaOH x LNaOH
The MNaOH is the average molarity of NaOH from Part I and the LNaOH is the volume of the base in each trial required to titrate the unknown in Part II.
As in Part I at the equivalence point
3 moles of NaOH = 1 mole of citric acid
mole citric acid in each trial = moles of NaOH x 1 mole citric acid3 mole NaOH
From here, we determine the mass of the citric acid in the unknown impure sample for each trial.
mass of citric acid in the unknown sample = mole citric acid x 192.124 g citric acid1 mol citric acid
To determine the mass % of the citric acid in the unknown we need to determine how much of the unknown was dissolved in the 20.0 mL we used in each trial. This is done with a simple ratio.
If we know that 0.900 g of the unknown sample was dissolved in 100.0 mL of solution, we can set up our equation. Therefore, if we used 20.0 mL of the solution in the trial, we can determine the g of unknown impure sample in the 20.0 mL as follows:
0.900 g unknown sample100.0 m L of solution = X g unknown sample in the trial20.0 mL of solution
Solving for X we see that 0.180 g is the mass of the unknown impure sample in the trial. If your measured amounts are different, use your measured numbers accordingly. We can now determine the mass % of citric acid for each trial.
mass % of citric acid in unknown = mass citric acid in unknown sample for each trialmass of unknown impure sample in each trial x 100%
average % of citric acid in the sample = Trial 1 + Trial 2 + Trial 33
Sample Problems:
- The student used 9.1 mL of a NaOH solution to titrate a 21.0 mL sample of a pure citric acid solution. The starting molarity of the citric acid solution was 0.014 M. What is the molarity of the NaOH solution?
mole of citric acid in the trial = Molaritycitric acid x Lcitric acid = (0.014 M)(0.0210 L)
= 2.94 x 10−4 mole of citric acid
mole NaOH in the trial = moles of citric acid x 3 mole NaOH1 mole citric acid
= 2.94 x 10−4 mole of citric acid x 3 mole NaOH1 mole citric acid
= 8.82 x 10−4 mole NaOH
molarity of NaOH in the trial = mol NaOH L of NaOH solution
= 0.000882 mol NaOH 0.0091 L of NaOH solution
= 0.097 M NaOH
- It took 10.5 mL of a 0.750 M NaOH solution to neutralize 25.0 mL of an impure sample of citric acid. The 25.0 mL titration sample had 0.767 g of the dissolved impure unknown. What is the mass % of citric acid in the sample?
H3C6H5O7 (aq) + 3NaOH (aq) → 3H2O (l) + Na3C6H5O7 (aq)
moles of NaOH = MNaOH x LNaOH
moles of NaOH = 0.750 mol/L x 0.0105 L = 0.007875 mol NaOH
3 moles of NaOH = 1 mole of citric acid
moles of citric acid = mole NaOH x 1 mole citric acid3 mole NaOH
moles of citric acid = 0.007875 mol NaOH x 1 mole citric acid3 mole NaOH
= .002625 moles citric acid
mass of citric acid in unknown sample = 0.002625 mol citric acid x 192.124 g citric acid1 mol citric acid
= 0.504 g citric acid
mass % of citric acid = 0.504 g0.767 g x 100%
= 65.7% citric acid in sample
Acid-Base Titration Lab Report Sheet
| Name: | Quiz is 20 points |
| Lab Report is 80 points |
Part I: Standardization of NaOH using Anhydrous Citric Acid
| Trial 1 | Trial 2 | Trial 3 | |
| Starting g of solid citric acid | 0.320 g | 0.331 g | 0.315 g |
| Starting M of citric acid solution | M | M | M |
| Volume of citric acid | 20.00 mL | 17.80 mL | 22.19 mL |
| Moles of citric acid | mol | mol | mol |
| Moles of NaOH | mol | mol | mol |
| Initial graduated cylinder volume | 10.00 mL | 10.00 mL | 10.00 mL |
| Final graduated cylinder volume | 5.20 mL | 5.50 mL | 4.70 mL |
| Volume of NaOH used | mL | mL | mL |
| Volume of NaOH used in L | L | L | L |
| NaOH concentration | M | M | M |
Average Molarity of NaOH ______________________
Show your work for one trial as well as the average molarity.
Part II: Determination of % Citric Acid in Unknown Impure Sample
| Trial 1 | Trial 2 | Trial 3 | |
| Volume unknown solution | 20.00 mL | 19.40 mL | 20.20 mL |
Mass of unknown impure sample (Remember 0.900 g are dissolved in 100.0 mL) | g | g | g |
| Initial graduated cylinder volume | 10.00 mL | 10.00 mL | 10.00 mL |
| Final graduated cylinder volume | 2.63 mL | 2.51 mL | 2.60 mL |
| Volume of NaOH used | mL | mL | mL |
| Volume of NaOH used in L | L | L | L |
| Moles of NaOH | mol | mol | mol |
| Moles of citric acid | mol | mol | mol |
| Mass of citric acid | g | g | g |
| % citric acid in a sample | % | % | % |
Average % citric acid in Unknown ________________
Show your work for one trial as well as the average % citric acid.
Conclusion: Write your conclusion here based on the Rubric for Grading Conclusions pdf posted to our online class.
Questions:
- Write the Molecular, Complete Ionic, and Net Ionic equations for the neutralization of lithium hydroxide with hydrobromic acid.
It took 10.40 mL of a NaOH solution to neutralize 0.307 g of KHP. What is the molarity of the NaOH solution? KHP means potassium hydrogen phthalate and has a molar mass of 204.2 g/mol.
- KHP (aq)+ NaOH (aq) → H2O (l) + NaKP (aq)
It took 8.82 mL of a 0.101 M NaOH solution to neutralize the KHP in a 0.501 g sample of impure KHP. What is the percentage of the KHP in the sample?

Corporate Finance A Focused Approach
ISBN: 978-1305637108
6th edition
Authors: Michael C. Ehrhardt, Eugene F. Brigham




