From the following information taken from the records of Peach Company at December 31 of this...
Fantastic news! We've Found the answer you've been seeking!
Question:
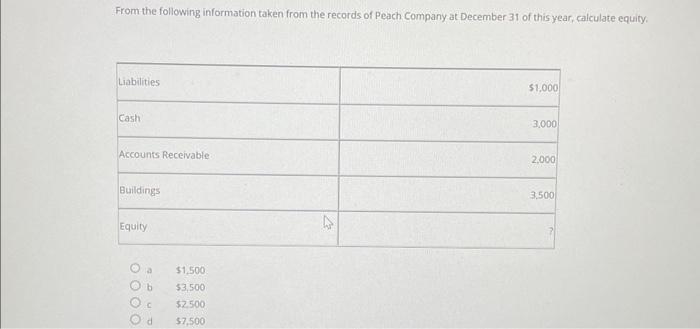
Transcribed Image Text:
From the following information taken from the records of Peach Company at December 31 of this year, calculate equity. Liabilities Cash Accounts Receivable Buildings Equity 0000 $1,500 b $3,500 C $2.500 d $7,500 4 $1,000 3,000 2,000 3,500 From the following information taken from the records of Peach Company at December 31 of this year, calculate equity. Liabilities Cash Accounts Receivable Buildings Equity 0000 $1,500 b $3,500 C $2.500 d $7,500 4 $1,000 3,000 2,000 3,500
Expert Answer:
Related Book For 

Accounting Tools for Business Decision Making
ISBN: 978-1118128169
5th edition
Authors: Paul D. Kimmel, Jerry J. Weygandt, Donald E. Kieso
Posted Date:
Students also viewed these accounting questions
-
The following information has been extracted from Lathoms books of account for the year to 30 April 2016: Required: (a) Prepare Lathoms trading account for the year to 30 April 2016. (b) State where...
-
On January 1, 2010, Cyber Company established a noncontributory defined benefit plan covering all employees and contributed $200,000 to the plan. At December 31, 2010, Cyber detemined that the 2010...
-
The Mago Company at December 31 has cash $50,000, noncash assets $250,000, liabilities $138,000, and the following capital balances: Gonzalez $112,000 and Maldonado $50,000. The firm is liquidated,...
-
Gothic Kings Ltd. Is a 100% owned subsidiary of Hadrian Inc. Gothic has been profitable in the past but incurred a loss for the year ended December 31, 20X3. Hadrian has indicated that if Gothic...
-
A satellite moves on a circular earth orbit that has a radius of 6.7 106 m. A model airplane is flying on a 15-m guideline in a horizontal circle. The guideline is parallel to the ground. Find the...
-
How has hiring women and minorities improved Xeroxs profitability ?
-
Six tests are performed, and the smallest P-value is 0.03. Which is the best conclusion? i. Reject H0 because P < 0.05. ii. Do not reject H0 because the Bonferroni adjustment yields P = 6(0.03) =...
-
Preparing a classified balance sheet Required Use the following information to prepare a classified balance sheet for Steller Co. at the end of 2012. Accounts receivable .... $42,500 Accounts payable...
-
Part II: Decision Analysis A customer has approached XYZ bank for a $50,000 one-year loan at 12% interest. If the bank does not approve the loan, the $50,000 will be invested in bonds that earn a 4%...
-
Crockett Corporation, a calendar year C corporation, reports net income of $2,000,000,000 on its 2023 audited financial statements (including financial statement depreciation of $250,000,000). Also,...
-
According to the historical data, the life expectancy in Mexico is equal to the life expectancy in Spain. A new study has been made to see whether this has changed. Records of 210 individuals from...
-
On 12 November 2013, Hazel made a gross chargeable transfer to a relevant property trust of 266,000 (after deduction of exemptions). On 1 April 2019 she gave 300,000 to her grandson. These were her...
-
Tony died on 11 July 2023, leaving an estate which was valued at 900,000. None of the transfers made on his death were exempt from IHT. He had made the following transfers during his lifetime:...
-
Tracey is a sole trader. She has the following transactions during the quarter to 31 December 2023 (all amounts shown are VAT-exclusive): Tracey drives a car with an emission rating of 118 g/km and...
-
On 7 August 2023, a VAT-registered company files its VAT return for the three months to 30 June 2023. This return shows that VAT of 18,200 is payable to HMRC, but the company does not pay this sum...
-
Smart Ltd began trading on 1 September 2023. Taxable turnover for the first four months of trading was as follows: On 1 November 2023, the company signed a contract to supply standard-rated services...
-
Use the reactions given below to determine AHxn for the following reaction: MN2()+3T2(g) MT2(g) +2NT2(g) Given: M(s)+ T2(g) MT2(g) AH -253.5 kJ N(s)+T2(g) NT2(g) AH -226.2 kJ M(s) + 2N(s) MN2(1) AH...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Boscan Corporation purchased machinery on January 1, 2014, at a cost of $250,000. The estimated useful life of the machinery is 4 years, with an estimated salvage value at the end of that period of...
-
On May 15, Criqui Outback Clothiers borrowed some money on a 4-month note to provide cash during the slow season of the year. The interest rate on the note was 8%. At the time the note was due, the...
-
In recent years, Farr Company has purchased three machines. Because of frequent employee turnover in the accounting department, a different accountant was in charge of selecting the depreciation...
-
What other examples can you imagine that might easily incorporate the Factory Method pattern?
-
What other examples can you imagine that might easily incorporate the Adapter pattern?
-
Why can you not label the static instance() method as virtual in Singleton and override it in President?

Study smarter with the SolutionInn App



