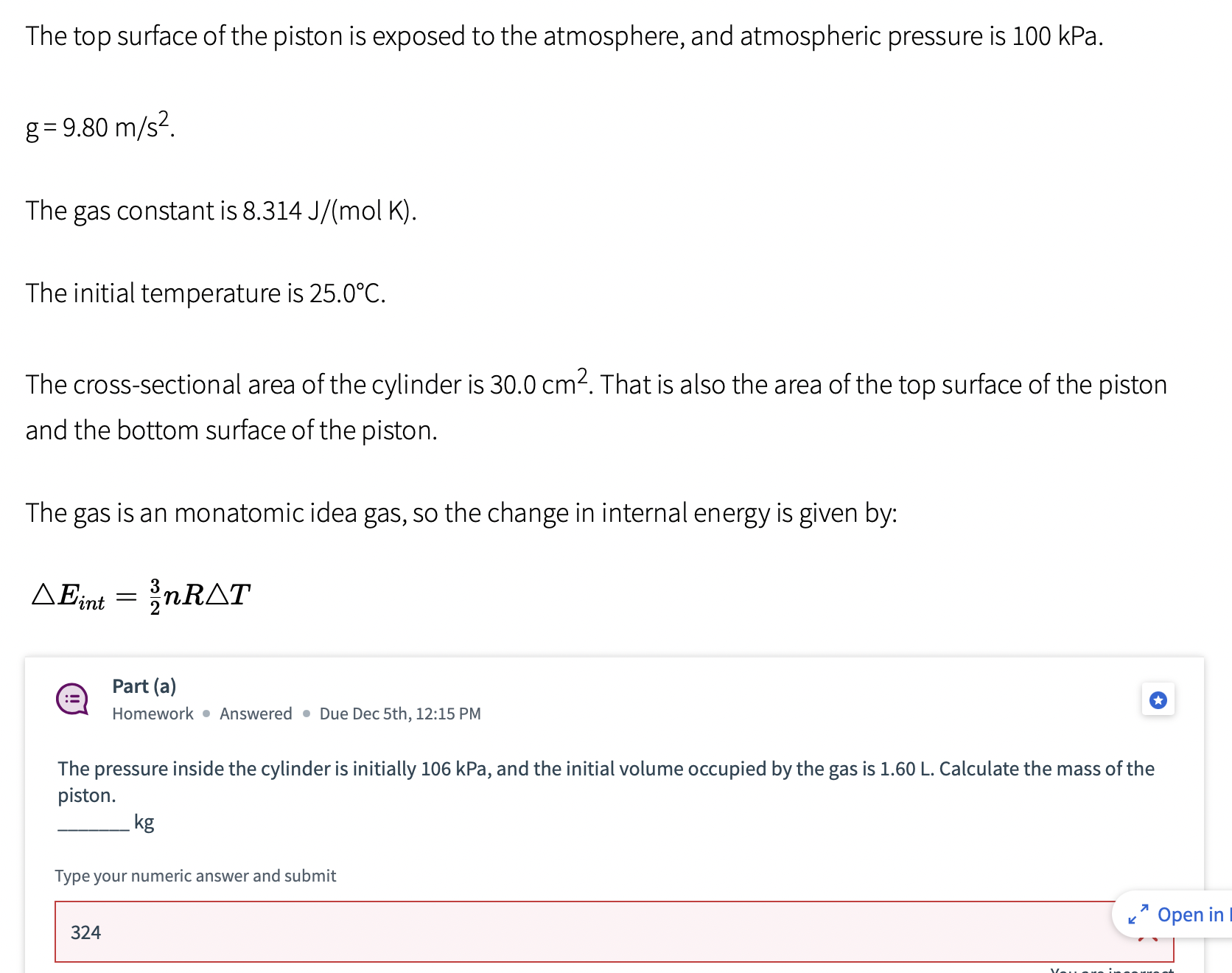
Gas in a cylinder Vi A cylinder of ideal gas is shown above. The gas is...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Gas in a cylinder Vi A cylinder of ideal gas is shown above. The gas is sealed in the region below the piston, and the piston is free to move up or down without friction. However, the piston starts in its equilibrium position. Use these numbers: The top surface of the piston is exposed to the atmosphere, and atmospheric pressure is 100 kPa. g= 9.80 m/s. The gas constant is 8.314 J/(mol K). K Open in Re The top surface of the piston is exposed to the atmosphere, and atmospheric pressure is 100 kPa. g= 9.80 m/s. The gas constant is 8.314 J/(mol K). The initial temperature is 25.0C. The cross-sectional area of the cylinder is 30.0 cm. That is also the area of the top surface of the piston and the bottom surface of the piston. The gas is an monatomic idea gas, so the change in internal energy is given by: AEint = nRAT := Part (a) Homework Answered Due Dec 5th, 12:15 PM The pressure inside the cylinder is initially 106 kPa, and the initial volume occupied by the gas is 1.60 L. Calculate the mass of the piston. kg Type your numeric answer and submit 324 7 Open in Part (b) Homework Unanswered Due Dec 5th, 12:15 PM Calculate the number of moles of ideal gas sealed in the cylinder. moles Type your numeric answer and submit Unanswered 5 attempts left Submit Part (c) Homework Unanswered Due Dec 5th, 12:15 PM Some heat is slowly added to the gas, so the gas expands at constant pressure to a final volume of 3.50 L. Calculate the work done by the gas inside the cylinder during this expansion. J Type your numeric answer and submit 7 Open in Reading Part (d) := Homework Unanswered Due Dec 5th, 12:15 PM Determine the final temperature of the gas, in Celsius. C Type your numeric answer and submit Unanswered 5 attempts left == Part (e) Homework Unanswered Due Dec 5th, 12:15 PM Determine how much heat was added to the gas. J Type your numeric answer and submit Submit + Open in Reading Gas in a cylinder Vi A cylinder of ideal gas is shown above. The gas is sealed in the region below the piston, and the piston is free to move up or down without friction. However, the piston starts in its equilibrium position. Use these numbers: The top surface of the piston is exposed to the atmosphere, and atmospheric pressure is 100 kPa. g= 9.80 m/s. The gas constant is 8.314 J/(mol K). K Open in Re The top surface of the piston is exposed to the atmosphere, and atmospheric pressure is 100 kPa. g= 9.80 m/s. The gas constant is 8.314 J/(mol K). The initial temperature is 25.0C. The cross-sectional area of the cylinder is 30.0 cm. That is also the area of the top surface of the piston and the bottom surface of the piston. The gas is an monatomic idea gas, so the change in internal energy is given by: AEint = nRAT := Part (a) Homework Answered Due Dec 5th, 12:15 PM The pressure inside the cylinder is initially 106 kPa, and the initial volume occupied by the gas is 1.60 L. Calculate the mass of the piston. kg Type your numeric answer and submit 324 7 Open in Part (b) Homework Unanswered Due Dec 5th, 12:15 PM Calculate the number of moles of ideal gas sealed in the cylinder. moles Type your numeric answer and submit Unanswered 5 attempts left Submit Part (c) Homework Unanswered Due Dec 5th, 12:15 PM Some heat is slowly added to the gas, so the gas expands at constant pressure to a final volume of 3.50 L. Calculate the work done by the gas inside the cylinder during this expansion. J Type your numeric answer and submit 7 Open in Reading Part (d) := Homework Unanswered Due Dec 5th, 12:15 PM Determine the final temperature of the gas, in Celsius. C Type your numeric answer and submit Unanswered 5 attempts left == Part (e) Homework Unanswered Due Dec 5th, 12:15 PM Determine how much heat was added to the gas. J Type your numeric answer and submit Submit + Open in Reading
Expert Answer:

Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Posted Date:
Students also viewed these physics questions
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
93) Clay of the Land is a manufacturer of glazed clay pots. Currently, it produces 300 clay pots per month which it sells through nurseries at a constant price of $5 per pot. Current demand for clay...
-
Consider the following discrete probability distribution: x P(x) 5..0.10 10..0.15 15..0.25 20..0.50 a. Calculate the expected value of the random variable. b. Let y = x + 5. Calculate the expected...
-
In Fig 12-16, if the frequency of the speakers were lowered, would the points D and C (where destructive and constructive interference occur) move farther apart or closer together?
-
Regal Freightway provides freight service. The companys balance sheet includes Land, Buildings, and Motor-Carrier Equipment. Regal Freightway uses a separate accumulated depreciation account for each...
-
The Hernandez Manufacturing Company has two producing departments, machining and assembly. Mr. Hernandez recently automated the machining department. The installation of a CAM system, together with...
-
BML Ltd. has an existing bond that trades on the secondary market. This bond has 11.5 years left to maturity, makes semiannual coupon payments, trades at a YTM of 10.25%, and has a current market...
-
The Fourth Amendment protects individuals from unreasonable searches and seizures and requires two branches of government to agree in order for search warrants to be issued. But what happens when the...
-
A group of new machines was purchased on February 17, 2021, under a royalty agreement with the following terms: The purchaser, Keller Corp., is to pay a royalty of $1 to the machinery supplier for...
-
Discount Pizza retires its 7% bonds for $68,000 before their scheduled maturity. At the time, the bonds have a face amount of $70,000 and a carrying value of $64,168. Record the early retirement of...
-
How do you think the use of slang words and phrases influences communication between English-speakers and people from other cultures? Or regional slang among different English-speaking dialects?...
-
A large fast-food restaurant is having a promotional game where game pieces can be found on various products. Customers can win food or cash prizes. According to the company, the probability of...
-
As general manager of the company Respira Feliz Ltd., which is dedicated to the manufacture of two different types of air cleaning systems in residential spaces: Singermen HTX235 and No bacterium...
-
We are passing in a list of numbers. Add up all the list elements and output the result. Iimport sys numbers = sys.argv[1].split(",") Expected output is 25
-
Let X be a random variable taking on values a1, a2, . . . , pr with probabilities p1, p2, . . . , pr and with E(X) = μ. Define the spread of X as follows: This, like the standard deviation, is a...
-
Explain the procedure for drawing the Mohrs circle under biaxial loading.
-
Explain the procedure for drawing Mohrs circle under complex stresses.
-
Derive an expression for normal and shear stresses in a body subjected to biaxial loading.

Study smarter with the SolutionInn App


