Gaseous hydrogen and oxygen are burned at latm under rich conditions: O2 + 5H2 aH20...
Fantastic news! We've Found the answer you've been seeking!
Question:
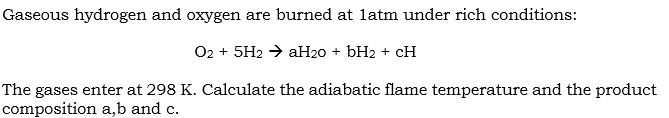
Transcribed Image Text:
Gaseous hydrogen and oxygen are burned at latm under rich conditions: O2 + 5H2 → aH20 + bH2 + CH The gases enter at 298 K. Calculate the adiabatic flame temperature and the product composition a,b and c. Gaseous hydrogen and oxygen are burned at latm under rich conditions: O2 + 5H2 → aH20 + bH2 + CH The gases enter at 298 K. Calculate the adiabatic flame temperature and the product composition a,b and c.
Expert Answer:
Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these chemical engineering questions
-
Refer to the circuit in the following figure 1. Select the correct approximate output signal from figure 2. Figure 1 2 k 30 V - 30 V 12 V (h) Figure 2 30 V A IK 12 V- - 12 V (a) (b) 12 V - 12 V 30 V...
-
Liquid hydrogen and oxygen are burned in a combustion chamber and fed through a rocket nozzle which exhausts at exit pressure equal to ambient pressure of 54 kPa. The nozzle exit diameter is 45 cm,...
-
Suppose hydrogen and oxygen are diffusing through air. A small amount of each is released simultaneously. How much time passes before the hydrogen is 1.00 s ahead of the oxygen? Such differences in...
-
The functions in Exercises 1128 are all one-to-one. For each function, a. Find an equation for f -1 (x), the inverse function. b. Verify that your equation is correct by showing that f( f -1 (x)) = x...
-
A corporation has 272 accounts receivable in a particular category. A random sample of 50 of them was taken. The sample mean was $492.36, and the sample standard deviation was $149.92. a. Find a 99%...
-
A water pump that consumes 2 kW of electric power when operating is claimed to take in water from a lake and pump it to a pool whose free surface is 30 m above the free surface of the lake at a rate...
-
Perform a thorough influential analysis of the air pollution and mortality data given in Table B.15. Perform any appropriate transformations. Discuss your results. City Mort Precip Educ Nonwhite Nox...
-
Laura Oil Company is considering investing in a new oil well. It is expected that the oil well will increase annual revenues by $130,000 and will increase annual expenses by $80,000 including...
-
Create an multiple step income statement following GAAP format and terminology. Assume 2 3 0 0 0 preferred stock shares and 4 4 0 0 0 common stock shares are outstanding for all 2 0 X 2 and a 2 0 %...
-
The following relate to auditors independence: a. Why is independence so essential for auditors? b. Compare the importance of independence of CPAs with that of other professionals, such as attorneys....
-
Chris Inc. will deposit $35,500 in a 5% fund at the end of each year for 10 years beginning December 31, 2017. What amount will be in the fund immediately after the last deposit?
-
Calculate the concentrations of each ion remaining in solution after precipitation is complete. Assume that AgCl is completely insoluble. Concentration of Ag* = ,M Concentration of NO. | MI 2+...
-
2. A car has an initial speed vo = 20 m/s. If it increases its speed along the circular track at s = 0, at = (0.8s) m/s, where s is in meters, determine the time needed for the car to travel s = 25 m.
-
The manufacturing company you work for installs a PLC control system on its assembly line, counting the number of components produced every shift. For quite a while, the system works without any...
-
1. How much money will you have in 8 years if you invest $4000 at 3.5% compounded quarterly? 2. How much money would you have after 7 years if you invested $12,000 at 2% in order to have compounded...
-
What is the crux of the ongoing discourse surrounding the efficacy of for-profit hospital models, particularly concerning whether the profit motive adversely affects the quality of care and...
-
What is the probability of having a Type I error with control limits set + 30 from the mean? O 0.26% O 2.6% O 3% O 4.56%
-
Hotel Majestic is interested in estimating fixed and variable costs so that the company can make more accurate projections of costs and profit. The hotel is in a resort area that is particularly busy...
-
A rigid tank contains 3.2 kg of refrigerant-134a initially at 26oC and 140 kPa. The refrigerant is now cooled until its pressure drops to 100 kPa. Determine (a) The entropy change of the refrigerant....
-
Determine the mass of one molecule of water (H2O). Assume the density to be 1000 kg/m3.
-
A four-cylinder two-stroke engine operating at 2000 rpm produces 50 kW of total brake power. If the cylinder displacement is 1 L, determine (a) The net work output in kJ per cylinder per cycle, (b)...
-
What is an organ system?
-
What is homeostasis?
-
During an action potential, (a) the membrane potential becomes more negative until it hits threshold. (b) the membrane potential increases as potassium ions flow out. (c) the membrane potential...

Study smarter with the SolutionInn App



