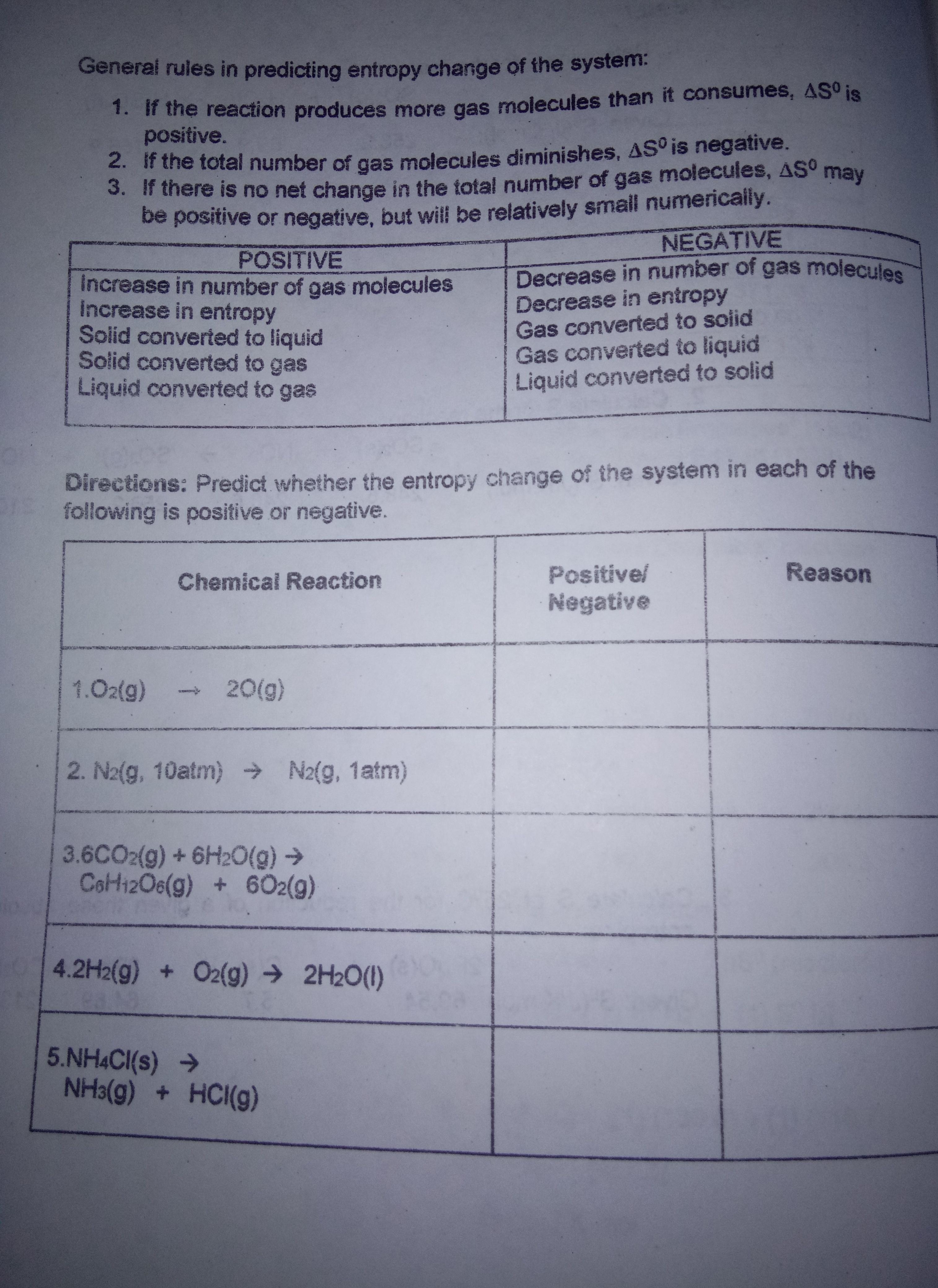
General rules in predicting entropy change of the system: 1. If the reaction produces more gas...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
General rules in predicting entropy change of the system: 1. If the reaction produces more gas molecules than it consumes, ASº is positive. 2. If the total number of gas molecules diminishes, AS° is negative. 3. If there is no net change in the total number of gas molecules, AS° may be positive or negative, but will be relatively small numerically. NEGATIVE POSITIVE Decrease in number of gas molecules Increase in number of gas molecules Increase in entropy Solid converted to liquid Solid converted to gas Liquid converted to gas Decrease in entropy Gas converted to solid Gas converted to liquid Liquid converted to solid Directions: Predict whether the entropy change of the system in each of the following is positive or negative. Chemical Reaction Positive/ Reason Negative 1.02(g) 20(g) 2. N2(g, 10atm) → N2(g, 1atm) 3.6CO2(g) +6H20(g) → CoH12O6(g) + 602(g) 4.2H2(g) + O2(g) → 2H2O(1) 5.NH4CI(s) > NH3(g) + HCI(g) General rules in predicting entropy change of the system: 1. If the reaction produces more gas molecules than it consumes, ASº is positive. 2. If the total number of gas molecules diminishes, AS° is negative. 3. If there is no net change in the total number of gas molecules, AS° may be positive or negative, but will be relatively small numerically. NEGATIVE POSITIVE Decrease in number of gas molecules Increase in number of gas molecules Increase in entropy Solid converted to liquid Solid converted to gas Liquid converted to gas Decrease in entropy Gas converted to solid Gas converted to liquid Liquid converted to solid Directions: Predict whether the entropy change of the system in each of the following is positive or negative. Chemical Reaction Positive/ Reason Negative 1.02(g) 20(g) 2. N2(g, 10atm) → N2(g, 1atm) 3.6CO2(g) +6H20(g) → CoH12O6(g) + 602(g) 4.2H2(g) + O2(g) → 2H2O(1) 5.NH4CI(s) > NH3(g) + HCI(g)
Expert Answer:
Answer rating: 100% (QA)
The entropy change positivenegative alongwith reasons is listed below SNo Entropy chan... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Predict the sign of the entropy change of the system for each of the following reactions: (a) N2(g) + 3 H2(g) 2 NH3(g) (b) CaCO3(s) CaO(s) + CO2(g) (c) 3 C2H2 (g) C6H6 (g) (d) Al2O3(s) + 3 H2(g) ...
-
Predict whether the entropy change is positive or negative for each of these reactions: (a) Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) (b) O(g) + O(g) O2(g) (c) NH4NO3(s) N2O(g) + 2H2O(g) (d) 2H2O2(l) ...
-
Predict whether unemployment will increase or decrease as a result of each of the following monetary policies. If it is unanticipated? What if it is anticipated? a. A reduction in the discount rate...
-
A function f: S is convex if and only if for all f (21x1 + a2x2 + ..+ x, Xn)
-
Let G be the volume (in gallons) of gasoline in a cars gas tank in which there are at most 5 gallons. Describe the volume of gasoline in the tank using inequality notation, interval notation, and a...
-
How should the servicescape of a firm that targets ethical shoppers be designed?
-
Diageo North America, Inc., the owner of the Bulleit brand family of whiskeys, sued competitor W.J. Deutsch & Sons Ltd. for allegedly infringing upon the trade dress of Diageos Bulleit bottles....
-
Doughboy Bakery would like to buy a new machine for putting icing and other toppings on pastries. These are now put on by hand. The machine that the bakery is considering costs $90,000 new. It would...
-
The security professional needs to be aware of the important security concepts that apply during software development and in the production environment. Which command in data control language...
-
Stang Corporation issued to Bradley Company $400,000 par value, 10-year bonds with a coupon rate of 12 percent on January 1, 20X5, at 105. The bonds pay interest semiannually on July 1 and January 1....
-
When time is an important constraint on a consumers choices, the consumers choice problem (when 3 activities x 1 , x 2 , and x 3 are chosen) is MaxU (x 1 , x 2 , x 3 ) + (M - p 1 x 1 - p 2 x 2 - p 3...
-
PunchTab Inc - venture capital (MDV) vs. Angel Investor. Which is the best investment?
-
Conditions: Imagine that you just started working for a financial organization that provides several financial calculators as web services that are available to third-party developers. Kelsey has...
-
Describing the relationships between the various financial statements. How does one transfer information to the other statements? What stories do they tell?
-
Define the two possible types of capital rationing, and discuss how capital rationing affects the attainment of management's goal of maximizing shareholders' value.
-
In which type of hedge fund investment, managers buy or sell securities and then hedge part or all of the associated risks? Question 7Answer Distressed securities Convertible arbitrage strategies...
-
The exchange rates in New York are: $1 = AUD 1.113 and $1 = 0.658 A dealer is offering a quote: AUD 1 = 0.7617. What is the profit you can earn on $24,541 using triangle arbitrage? You are given the...
-
Define the essential properties of the following types of operating systems: a. Batch b. Interactive c. Time sharing d. Real time e. Network f. Parallel g. Distributed h. Clustered i. Handheld
-
An unknown salt is either NaF,NaCl, or NaOCl. When 0.050 mol of the salt is dissolved in water to form 0.500 L of solution, the pH of the solution is 8.08.What is the identity of the salt?
-
Draw the structural formulas for three isomers of pentane, C5H12.
-
Identify each of the following as measurements of length, area, volume, mass, density, time, or temperature: (a) 25 ps, (b) 374.2 mg, (c) 77 K, (d) 100,000 km2 (e) 1.06 m, (f) 16 nm2, (g) -78 C, (h)...
-
a. The yield to maturity on two 10-year maturity bonds currently is 7%. Each bond has a call price of $1,100. One bond has a coupon rate of 6%, the other 8%. Assume for simplicity that bonds are...
-
Suppose we add a new variable equal to current liabilities/current assets to Altmans equation. Would you expect this variable to receive a positive or negative coefficient?
-
On May 30, 2020, Janice Kerr is considering one of the newly issued 10-year AAA corporate bonds shown in the following exhibit. a. Suppose that market interest rates decline by 100 basis points...

Study smarter with the SolutionInn App