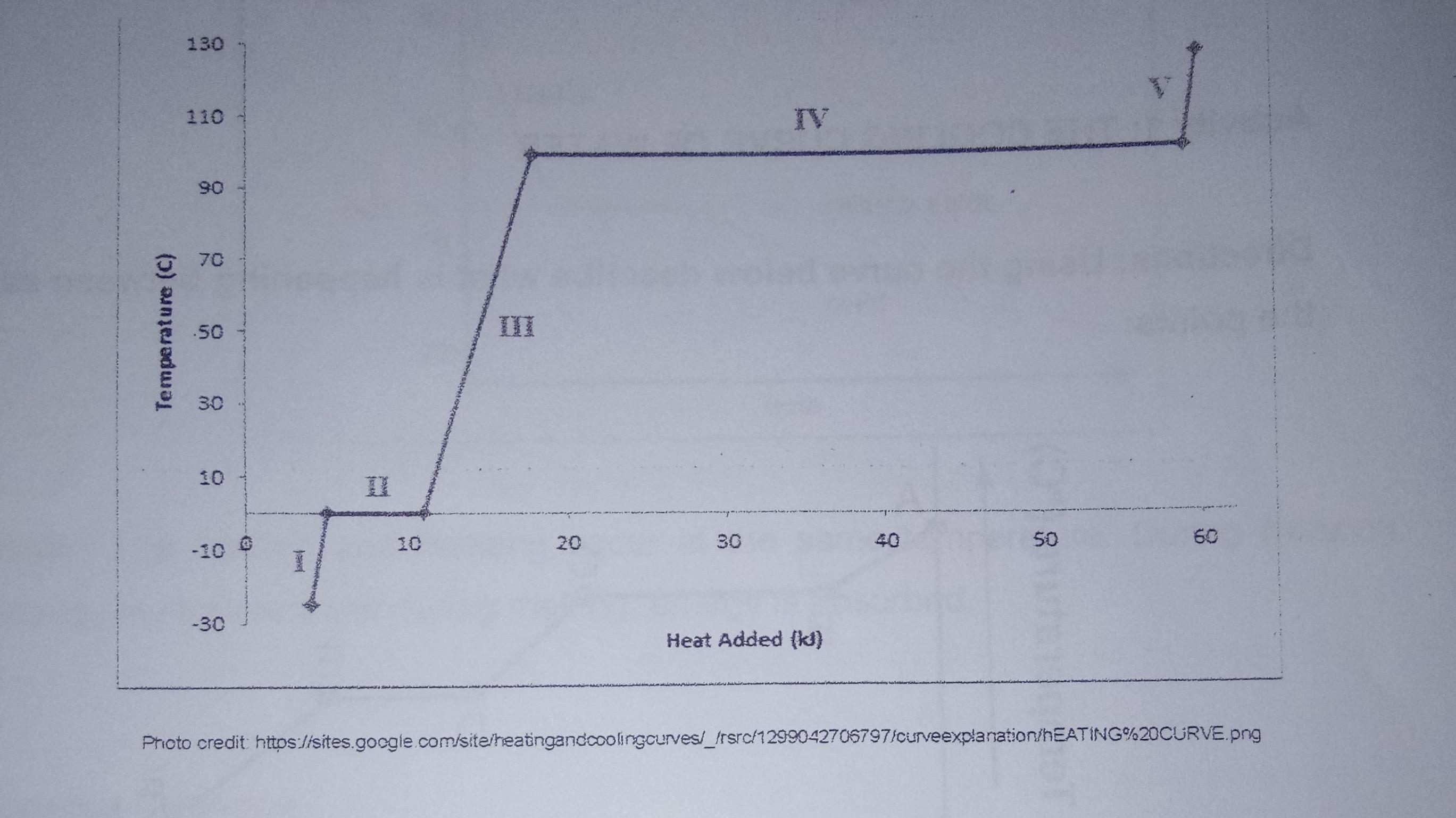
Use the cooling curve below to answer the following questions. 130 110 IV 06 70 .50...
Fantastic news! We've Found the answer you've been seeking!
Question:
![]()


Transcribed Image Text:
Use the cooling curve below to answer the following questions. 130 110 IV 06 70 .50 II 30 10 II 10 20 30 40 50 60 -10 -30 Heat Added (k) Photo credit https://sites.google.com/site/heatingandcoolingcurves/_frsrc/1299042706797/curveexplanation/hEATING%20CURVE.png Temperature (C) 1. In which region(s) does temperature remain constant? 2. In which region(s) does temperature increase? 3. In which region(s) of the graph does a phase change occur? 4. In which region(s) of the graph would the substance only be in one phase? 5. In which region(s) of the graph would the substance be a solid only? 6. In which region(s) of the graph would the substance be a solid and a liquid? 7. In which region(s) of the graph would the substance be a liquid and a gas? 8. In which region(s) of the graph would the substance be a gas only? 9. In which region(s) of the graph does boiling take place? 10. In which region(s) of the graph does melting take place? Use the cooling curve below to answer the following questions. 130 110 IV 06 70 .50 II 30 10 II 10 20 30 40 50 60 -10 -30 Heat Added (k) Photo credit https://sites.google.com/site/heatingandcoolingcurves/_frsrc/1299042706797/curveexplanation/hEATING%20CURVE.png Temperature (C) 1. In which region(s) does temperature remain constant? 2. In which region(s) does temperature increase? 3. In which region(s) of the graph does a phase change occur? 4. In which region(s) of the graph would the substance only be in one phase? 5. In which region(s) of the graph would the substance be a solid only? 6. In which region(s) of the graph would the substance be a solid and a liquid? 7. In which region(s) of the graph would the substance be a liquid and a gas? 8. In which region(s) of the graph would the substance be a gas only? 9. In which region(s) of the graph does boiling take place? 10. In which region(s) of the graph does melting take place?
Expert Answer:
Answer rating: 100% (QA)
Based on the diagram above we have the answers 1 Tem... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Use the heatingcooling curve below to answer the following questions. a. What is the freezing point of the liquid? b. What is the boiling point of the liquid? c. Which is greater, the heat of fusion...
-
Use the total cost curve below to answer the following questions. a. For what values of output is marginal product of labor decreasing? What is true about marginal cost over this range of output? b....
-
Use the information provided below to answer the following questions. Other information: ¢ All of the shares were issued during the first year of operations (year ended October 31, 2013). ¢...
-
The jet plane starts from rest at s = 0 and is subjected to the acceleration shown. Determine the speed of the plane when it has traveled 1000 ft. Also, how much time is required for it to travel...
-
Interview a production manager at a nearby goods-producing company to determine how the company plans its production for fluctuating demand. What approaches does the company use?
-
Describe four recommendations made by the National Commission on Fraudulent Financial Reporting.
-
Match the accounting term with the corresponding definition. 1. Accrual basis accounting 2. Matching principle 3. Revenue recognition principle 4. Fiscal period a. Any consecutive 12-month period. b....
-
Exhibit 3 2 contains a partial audit program for substantive tests of accounts receivable. Required: For audit procedures 1 4, identify the primary assertion being tested.
-
During the year, TRC Corporation has the following inventory transactions. Date Transaction Number of Units Unit Cost Total Cost January 1 Beginning inventory 47 $39 $1,833 April 7 Purchase 127 41...
-
Using the Public MACRO BITCOIN scorecard spreadsheet (linked in its associated masterclass lesson - Long Term 32), create a COPY of it and perform a complete analysis for the date 22/2/2022....
-
What is the value of egg_count after running this code? egg_count = 0 def buy_eggs(): egg_count += 12 buy_eggs()
-
Explain why system is tested for impulse and step inputs.
-
The following data are taken from the comparative balance sheets of Cascade Billiards Club, which prepares its financial statements using the accrual basis of accounting. Members are billed based...
-
A triangular pulse signal \(x(t)\) is shown in Fig. 1.77a. Sketch the following signals. (a) \(x(4 t)\); (b) \(x(4 t+3)\); (c) \(x(-3 t+2)\); (d) \(x\left(\frac{t}{3}+2ight)\); (e) \(x(3 t-2)\); (f)...
-
Action Quest Games Inc. adjusts its accounts annually. The following information is available for the year ended December 31, 2019. 1. Purchased a 1-year insurance policy on June 1 for $1,800 cash....
-
Southern Californian Scott Robinson had quite a rsum when he returned from studying in France last Christmas. The 26-year-old had an undergraduate degree from Bucknell University, two MBAs, and...
-
Remainder person R owned a contingent remained in a trust, which R transferred to R's child. Under the trust terms, R's sibling S was to receive the remainder if S survived the income beneficiary,...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
Balance each of the following oxidation reduction reactions by using the oxidation states method. a. C2H6(g) + O2(g) CO2(g) + H2O(g) b. Mg(s) + HCl(aq) Mg2+(aq) + Cl2(aq) + H2(g) c. Cu(s) + Ag+(aq)...
-
Calculate G o and K at 25 o C for the galvanic cell reactions in Exercise 21. Reactions in exercise 21. a. Cl 2 + 2e - 2Cl 2 E cell = 1.36 V Br 2 + 2e - 2Br 2 E cell = 1.09 V b. MnO 4 - + 8H + + 5e...
-
A sample of nitrogen gas was collected over water at 20. o C and a total pressure of 1.00 atm. A total volume of 2.50 10 2 mL was collected. What mass of nitrogen was collected? (At 20. o C the...
-
Determine the fundamental frequency of a uniform fixed-fixed beam carrying a mass \(M\) at the middle by applying Rayleigh's method. Use the static deflection curve for \(W(x)\).
-
Applying Rayleigh's method, determine the fundamental frequency of a cantilever beam (fixed at \(x=l\) ) whose cross-sectional area \(A(x)\) and moment of inertia \(I(x)\) vary as \(A(x)=A_{0} x /...
-
Using Rayleigh's method, estimate the fundamental frequency for the lateral vibration of a uniform beam fixed at both the ends. Assume the deflection curve to be \[W(x)=c_{1}\left(1-\cos \frac{2 \pi...

Study smarter with the SolutionInn App


