Given that the concentration of hydronium ions in a solution with pH of 2 is 0.19%,...
Fantastic news! We've Found the answer you've been seeking!
Question:

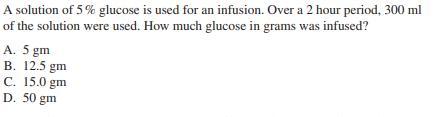
Transcribed Image Text:
Given that the concentration of hydronium ions in a solution with pH of 2 is 0.19%, what would be the concentration of hydronium ions in a solution with pH of 3? A. 0.13% B. 0.29% C. 0.019% D. 1.9% A solution of 5% glucose is used for an infusion. Over a 2 hour period, 300 ml of the solution were used. How much glucose in grams was infused? A. 5 gm B. 12.5 gm C. 15.0 gm D. 50 gm Given that the concentration of hydronium ions in a solution with pH of 2 is 0.19%, what would be the concentration of hydronium ions in a solution with pH of 3? A. 0.13% B. 0.29% C. 0.019% D. 1.9% A solution of 5% glucose is used for an infusion. Over a 2 hour period, 300 ml of the solution were used. How much glucose in grams was infused? A. 5 gm B. 12.5 gm C. 15.0 gm D. 50 gm
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Part 1 Hydronium ion concentration Firs... View the full answer

Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Posted Date:
Students also viewed these medical sciences questions
-
What is the concentration of hydronium ions in a solution that has a pH of 3? Why is such a solution impossible to prepare?
-
Sometimes the pH must be converted into the hydronium ion concentration. The quickest way to find the hydronium ion concentration in a solution is to use a pH meter to measure the pH and then...
-
Do economists have any particular expertise at making normative arguments? In other words, they have expertise at making positive statements (i.e., what will happen) about some economic policy, for...
-
Michael Porter argued that in many industries, "strategies converge and competition becomes a series of races down identical paths that no one can win." Briefly explain whether firms in these...
-
The trial balance for Hanna Resort Limited on August 31 is as follows: Additional information: 1. The balance in Prepaid Rent includes payment of the final month's rent and rent for August 2014. 2....
-
An indirect way of measuring of secondary emission from ponds or large bodies of water used in waste treatment is to measure the concentration and velocity over the surface. The data can then be...
-
Consider Wal-Mart, a large retailer. Classify the following items as an Asset (A), a Liability (L), or Stockholders Equity (S) for Wal-Mart: a . _____ Accounts payable b. _____ Common stock c. _____...
-
Use the exact interest method (365 days) and the ordinary interest method (360 days) to compare the amount (in $) of interest for the loan. (Round your answers to two decimal places.) Principal Rate...
-
You are the manager of the Mighty Fine mutual fund. The following table reflects the activity of the fund during the last quarter. The fund started the quarter on January 1 with a balance of $40...
-
An antisymmetric [0/90] laminate is loaded under a biaxial load Nx = Ny = No. The following strains and curvatures are obtained &=&=&o, Kx = -Ky = Ko. Assuming that Vy < < 1, obtain an expression for...
-
13. AN aircraft fittings use flare cones that are OA. 70 degrees. B. 45 degrees. OC. 55 degrees. O D. 37 degrees. Mark for review (Will be highlighted on the review page)
-
There are three types of materials with the properties shown below: Material #1 oy = 400 MPa E = 70 GPa p = 2.7 ton/m Material #2 oy = 750 MPa E = 210 GPa p = 7.8 ton/m Material #3 oy = 920 MPa E =...
-
Explain whether the thrust of a fixed pitch propeller changes with the flight speed and in what way. You may wish to use sketches to illustrate your explanation.
-
A three-blade propeller of a diameter of 1.5 m has a rotational speed is 60 Hz, the activity factor AF is 200 and its static torque coefficient is 0.05. The propellers integrated lift coefficient is...
-
A large crankshaft made of cast iron with fracture toughness (Kc = 50 MNm-3/2) undergoes cyclic tensile stress, 250 MPa, and compressive stress, 80 MPa. Before using the material, an inspection with...
-
(b) In November and December 2014, Lane Co., a newly organized magazine publisher, received cash of $60,000 for 1,000 three-year subscriptions at $20 per year, starting with the January 2015 Issue....
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Who has the shorter wavelength when running at the same speed: a person weighing 60 kg or a person weighing 80 kg? Explain your reasoning.
-
(a) A flask of volume 350. mL contains 0.1500 mol Ar at 24 C. What is the pressure of the gas in kilopascals? (b) You are told that 23.9 mg of bromine trifluoride exerts a pressure of 10.0 Torr at...
-
The chemists who synthesized the pentamethylcyclopentadienyl cation (see Exercise 11.11) found that its p-electrons are found in one double bond and a p-orbital that is delocalized across the three...
-
Under IFRS, income statements are required to show: A. Extraordinary items. B. Unusual items. C. Exceptional items. D. None of the above.
-
Set out in Figure 16.10 are summarized balance sheets and income statements for F Co. for 20X1 and 20X2. You are required to: Figure 16.10 a. prepare a table of ratios, covering all aspects of...
-
Earnings is calculated deducting: A. Dividends on ordinary shares. B. Dividends on preference shares. C. Tax expense. D. Interest expense.

Study smarter with the SolutionInn App


