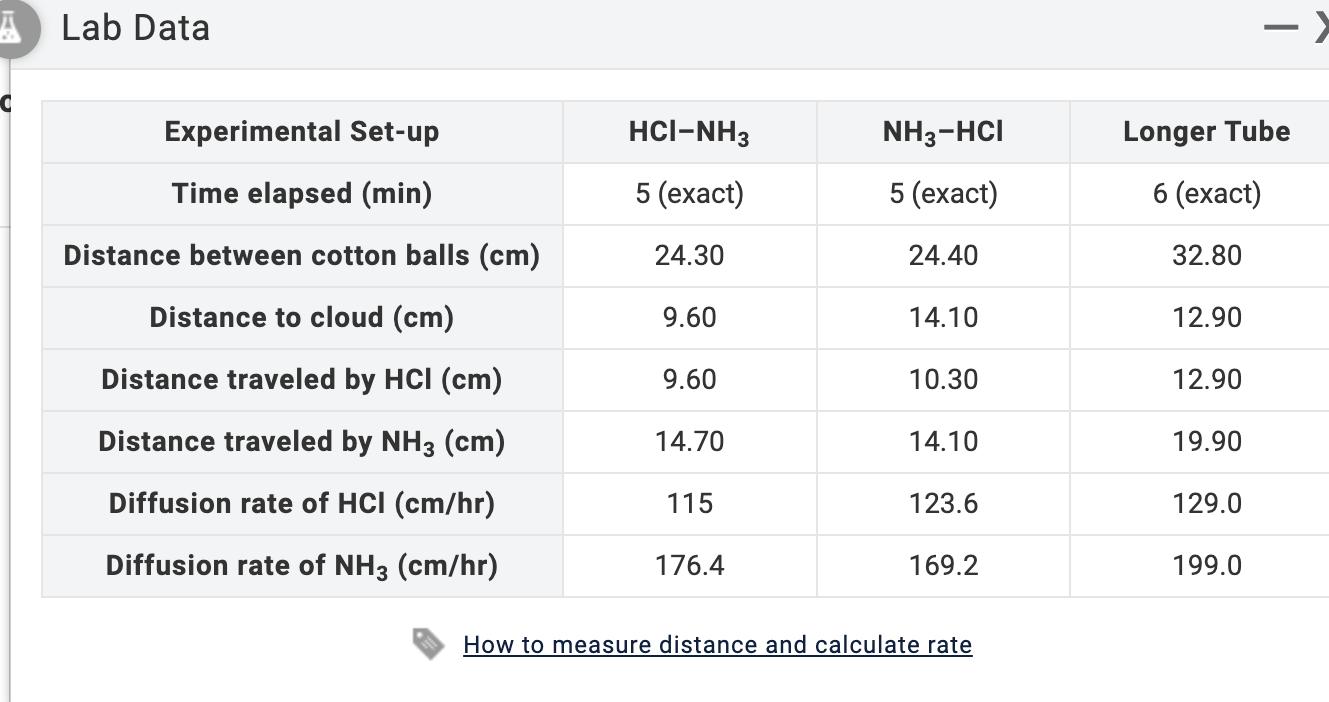
GAS LAW DIFFUSION AND GRAHAM'S LAW . INTRODUCTION LABORATORY SIMULATION 1) Given that the molar mass...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
GAS LAW DIFFUSION AND GRAHAM'S LAW . INTRODUCTION LABORATORY SIMULATION 1) Given that the molar mass of ammonia is 17.030 g/mol, calculate the molar mass of hydrogen chloride using your Phase 5 (Longer Tube) Lab Data. Molar mass of HCI = g/mol ALab Data C Experimental Set-up Time elapsed (min) Distance between cotton balls (cm) Distance to cloud (cm) Distance traveled by HCI (cm) Distance traveled by NH3 (cm) Diffusion rate of HCI (cm/hr) Diffusion rate of NH3 (cm/hr) HCI-NH3 5 (exact) 24.30 9.60 9.60 14.70 115 176.4 NH3-HCI 5 (exact) 24.40 14.10 10.30 14.10 123.6 169.2 How to measure distance and calculate rate Longer Tube 6 (exact) 32.80 12.90 12.90 19.90 129.0 199.0 GAS LAW DIFFUSION AND GRAHAM'S LAW . INTRODUCTION LABORATORY SIMULATION 1) Given that the molar mass of ammonia is 17.030 g/mol, calculate the molar mass of hydrogen chloride using your Phase 5 (Longer Tube) Lab Data. Molar mass of HCI = g/mol ALab Data C Experimental Set-up Time elapsed (min) Distance between cotton balls (cm) Distance to cloud (cm) Distance traveled by HCI (cm) Distance traveled by NH3 (cm) Diffusion rate of HCI (cm/hr) Diffusion rate of NH3 (cm/hr) HCI-NH3 5 (exact) 24.30 9.60 9.60 14.70 115 176.4 NH3-HCI 5 (exact) 24.40 14.10 10.30 14.10 123.6 169.2 How to measure distance and calculate rate Longer Tube 6 (exact) 32.80 12.90 12.90 19.90 129.0 199.0
Expert Answer:
Answer rating: 100% (QA)
By using Grahams law of diffusion the molar mass of a substance can be calculated as follows Rate 1 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is a SWOT analysis and why is it important to managers?
-
What is a SWOT analysis and why is it important?
-
What are strategic competitiveness, strategy, competitive advantage, above-average returns, and the strategic management process?
-
What were the reasons for the failure of the original purpose of FordDirect.com? Who eventually gained from the system and what were the gains?
-
Gloria has just made her ninth annual $2000 contribution to her RRSP. She now plans to make semiannual contributions of $2000. The first contribution will be made six months from now. How much will...
-
A proximity probe has a useful frequency range related to what operating parameter?
-
The number of A grades received in different subjects per student (a) find the mean, variance, and standard deviation of the probability distribution, (b) interpret the results. Grades 0 1 2 3 4 5 6...
-
1. Consider two firms engaging in sequential Stackelberg competition. Suppose firm 1 decides its quantity x1 first and firms 2 follows after observing x1. The demand function of the market is x(p) =...
-
The Final Report of the Joint Union/Management Task Force on Diversity and Inclusion makes 28 recommendations. Which one(s) do you find most important? Which organization(s) do you consider essential...
-
A company manufactures a single product. Budget and standard cost details for next year include: Selling price per unit ................................$24.00 Variable production cost per unit...
-
Brendan and Jisoo are shareholders in BJ, Inc., which has elected to be taxed as a C-corp. Brendan and Jisoo each own a 50% interest in BJ, Inc. Brendans basis in his stock is $38,000. Jisoos basis...
-
For problems 1-3, find the derivative of f(x). 1. f(x)=12x - 4x +5x-21x-5x+23 2. f(x)=12x7-4x4 +5x-21x5x +23 3 ((x) = -7/7 - 7 + 8 /x 3. f(x)=- 4. Find the coordinates of points where the graph of...
-
The kicker and the goalie play the penalty kick described above. Numbers (a,b) in the table are the kicker's payoff and the goalie's payoff, respectively. Select the correct answer from the choices...
-
Given these competing perspectives, do you see the JOBS act as overall positive or negative ?
-
As an insurance agent, how can I handle increases from natural disasters? Will it have a regional impact or a general growth?
-
A number of Daimler's former senior executives, who operated ina decentralized corporate structure, per-although Germany outlawed bribery in mitted or were directly involved in the 1999 when it...
-
(14%) Problem 1: In the 2012 remake of Total Recall, a plot device centers around an elevator that travels through the Earth powered solely by the force of gravity. For simplicity, suppose that the...
-
Suppose you need to answer any four of seven essay questions on a history test and you can answer them in any order. a. How many different question combinations are possible? b. What is the...
-
Describe four methods managers can use to acquire information about actual work performance.
-
How could you use the concept of control in your personal life? Be specific. (Think in terms of feedforward, concurrent, and feedback controls as well as specific controls for the different aspects...
-
Do followers make a difference in whether a leader is effective? Discuss.
-
Using a financial calculator, solve for the unknowns in each of the following situations. a. On June 1, 2024, Holly Golightly purchases lakefront property from her neighbor, George Peppard, and...
-
Sally W. Emanual, a teacher, had the following dividends and interest during 2022: Additional information pertaining to Sally Emanual includes The taxable portion of the pension is \($7,000.\) Sally...
-
Ed owns Oak Knoll Apartments. During the year, Fred, a tenant, moved to another state. Fred paid Ed \($1,000\) to cancel the two-year lease he had signed. Ed subsequently began renting the unit to...

Study smarter with the SolutionInn App


