Given these reactions, where X represents a generic metal or metalloid, 1) H2(g)+12O2(g)?H2O(g)?????1=?241.8 kJ 2) X(s)+2Cl2(g)?XCl4(s)?????2=+278.7 kJ
Fantastic news! We've Found the answer you've been seeking!
Question:
Given these reactions, where X represents a generic metal or metalloid, 1) H2(g)+12O2(g)?H2O(g)?????1=?241.8 kJ 2) X(s)+2Cl2(g)?XCl4(s)?????2=+278.7 kJ 3) 12H2(g)+12Cl2(g)?HCl(g)?????3=?92.3 kJ 4) X(s)+O2(g)?XO2(s)?????4=?747.7 kJ 5) H2O(g)?H2O(l)?????5=?44.0 kJ what is the enthalpy, ?H, for this reaction? XCl4(s)+2H2O(l)?XO2(s)+4HCl(g)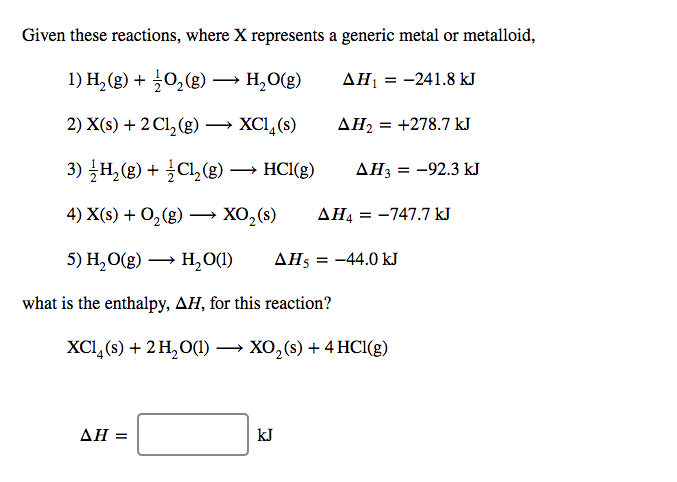
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:




