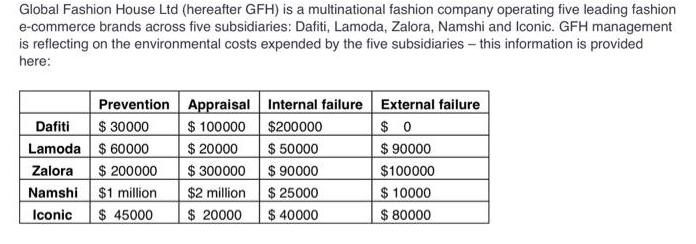
Global Fashion House Ltd (hereafter GFH) is a multinational fashion company operating five leading fashion e-commerce...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
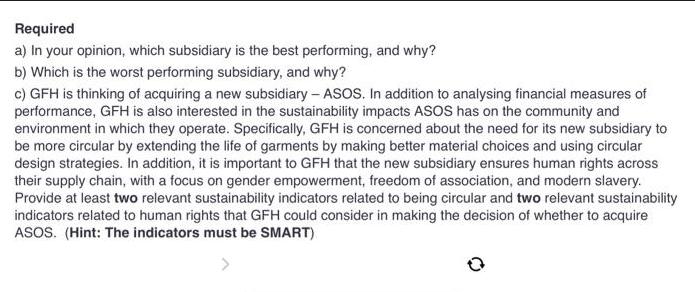
Global Fashion House Ltd (hereafter GFH) is a multinational fashion company operating five leading fashion e-commerce brands across five subsidiaries: Dafiti, Lamoda, Zalora, Namshi and Iconic. GFH management is reflecting on the environmental costs expended by the five subsidiaries - this information is provided here: Prevention Appraisal Internal failure $100000 $200000 $20000 $ 50000 $ 90000 $ 25000 $ 40000 Dafiti $ 30000 Lamoda $60000 Zalora Namshi Iconic $ 200000 $1 million $ 45000 $ 300000 $2 million $20000 External failure $0 $ 90000 $100000 $ 10000 $ 80000 Required a) In your opinion, which subsidiary is the best performing, and why? b) Which is the worst performing subsidiary, and why? c) GFH is thinking of acquiring a new subsidiary - ASOS. In addition to analysing financial measures of performance, GFH is also interested in the sustainability impacts ASOS has on the community and environment in which they operate. Specifically, GFH is concerned about the need for its new subsidiary to be more circular by extending the life of garments by making better material choices and using circular design strategies. In addition, it is important to GFH that the new subsidiary ensures human rights across their supply chain, with a focus on gender empowerment, freedom of association, and modern slavery. Provide at least two relevant sustainability indicators related to being circular and two relevant sustainability indicators related to human rights that GFH could consider in making the decision of whether to acquire ASOS. (Hint: The indicators must be SMART). > Global Fashion House Ltd (hereafter GFH) is a multinational fashion company operating five leading fashion e-commerce brands across five subsidiaries: Dafiti, Lamoda, Zalora, Namshi and Iconic. GFH management is reflecting on the environmental costs expended by the five subsidiaries - this information is provided here: Prevention Appraisal Internal failure $100000 $200000 $20000 $ 50000 $ 90000 $ 25000 $ 40000 Dafiti $ 30000 Lamoda $60000 Zalora Namshi Iconic $ 200000 $1 million $ 45000 $ 300000 $2 million $20000 External failure $0 $ 90000 $100000 $ 10000 $ 80000 Required a) In your opinion, which subsidiary is the best performing, and why? b) Which is the worst performing subsidiary, and why? c) GFH is thinking of acquiring a new subsidiary - ASOS. In addition to analysing financial measures of performance, GFH is also interested in the sustainability impacts ASOS has on the community and environment in which they operate. Specifically, GFH is concerned about the need for its new subsidiary to be more circular by extending the life of garments by making better material choices and using circular design strategies. In addition, it is important to GFH that the new subsidiary ensures human rights across their supply chain, with a focus on gender empowerment, freedom of association, and modern slavery. Provide at least two relevant sustainability indicators related to being circular and two relevant sustainability indicators related to human rights that GFH could consider in making the decision of whether to acquire ASOS. (Hint: The indicators must be SMART). >
Expert Answer:
Answer rating: 100% (QA)
a To determine which subsidiary is the best performing we can consider the overall environmental costs incurred by each subsidiary By summing up the costs under each category prevention appraisal inte... View the full answer

Related Book For 

Strategic Management Text and Cases
ISBN: 978-1259900457
9th edition
Authors: Gregory G Dess Dr., Gerry McNamara, Alan Eisner
Posted Date:
Students also viewed these accounting questions
-
The angular velocity and the amplitude of a simple pendulum is and a respectively. At a displacement x from the mean position if its kinetic energy is T and potential energy is V, then the ratio of T...
-
Subject : Strategic Management in a Global Environment Safaricom: Innovative Telecom Solutions to Empower Kenyans As the largest mobile provider in Kenya, Safaricom has touched the lives of Kenyans...
-
Iona Company, a large printing company, is in its fourth year of a five-year, quality improvement program. The program began in 2009 with an internal study that revealed the quality costs being...
-
Define a passive portfolio construction strategy and briefly discuss how reasonable it might be for an investor to pursue a passive strategy. Explain the role of risk aversion in the construction of...
-
A piston/cylinder arrangement has the piston loaded with outside atmospheric pressure and the piston mass to a pressure of 20 lbf/in 2, shown in Fig P5.28. It contains water at 25 F, which is then...
-
Nobo Uematsu Company has a balance in its Accounts Payable control account of \($8,250\) on January 1, 2008. The subsidiary ledger contains three accounts: Jones Company, balance \($3,000;\) Brown...
-
For the data in Exercise 7: a. Compute the expected frequencies for testing H0 : p1 = 0.4, p2 = 0.3, p3 = 0.1, p4 = 0.15, p5 = 0.05. b. Is it appropriate to perform a chi-square test for the...
-
State the effect (cash receipt or payment and amount) of each of the following transactions, considered individually, on cash flows: a. Sold 5,000 shares of $30 par common stock for $45 per share. b....
-
What effect did the Supreme Court's ruling in Shelby County v. Holder (2013) have in many states previously required to get preclearance like Texas, Alabama, and Georgia? Question 49 options: Many of...
-
Estimate the volume change of vaporization for ammonia at 20(C. At this temperature the vapor pressure of ammonia is 857 kPa.
-
7. A bond has face value 100 and annual coupons of size 6. The redemption value after 8 years is 125. Find the price of the bond if the annual yield rate is 8%
-
Welch Corporation just paid its annual dividend of $2.00 per share. The firm is expected to grow at a rate of 15 percent for the next three years and then at 6 percent per year thereafter. The...
-
Mary was so excited about the holiday shopping season that she could hardly wait for the Black Friday sales. She got up early and spent all day at her favorite store, Mega Deals, shopping. Mary was...
-
The following diagram represents the ownership of issued share capital of the companies in a group. What is the total non-controlling interest in D Ltd? A: 20 per cent B: 33.6 per cent C: 60 per...
-
A U.S. investor is considering investing $100 million in Fiat stock in Italy. The stock has an expected return of 5% in euros, including the dividend yield, and the investor is forecasting a 3%...
-
Express in terms of 1. -343 - 343 = (Simplify your answer. Type your answer in the form a + bi.)
-
Sealand Construction entered into a contract to construct a floating bridge across a lake. The contract price for the bridge is P7,500,000. During 2009, costs of P1,800,000 were incurred representing...
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
What unique responsibilities does United Way have to its stakeholders , and how successful has the organization been with its overall strategy? Is United Ways mission still viable? In 2017, United...
-
How should leadership manage innovation in this industry, and what are Weight Watchers options for growth? Weight Watchers International Inc. had been a major player in the weight-loss industry for...
-
What is the structure of the beer industry, and how does this affect choice of strategy? The Boston Beer Company, known for its Samuel Adams brand, is the largest craft brewery in the United States,...
-
Calculate the change in entropy for the conversion of \(1 \mathrm{~mol}\) of ice to liquid at \(273 \mathrm{~K}\) and \(1 \mathrm{~atm}\). The latent heat of fusion is \(6500 \mathrm{~J} /...
-
Calculate the change in entropy when 5 moles of an ideal gas expands from a volume of \(5 \mathrm{~L}\) to \(50 \mathrm{~L}\) at \(27^{\circ} \mathrm{C}\).
-
Calculate the entropy change when \(96 \mathrm{~g}\) of methane is heated from \(35^{\circ} \mathrm{C}\) to \(200^{\circ} \mathrm{C}\) at constant volume. Assume \(C_{V}=1.735 \mathrm{~kJ} /...

Study smarter with the SolutionInn App


