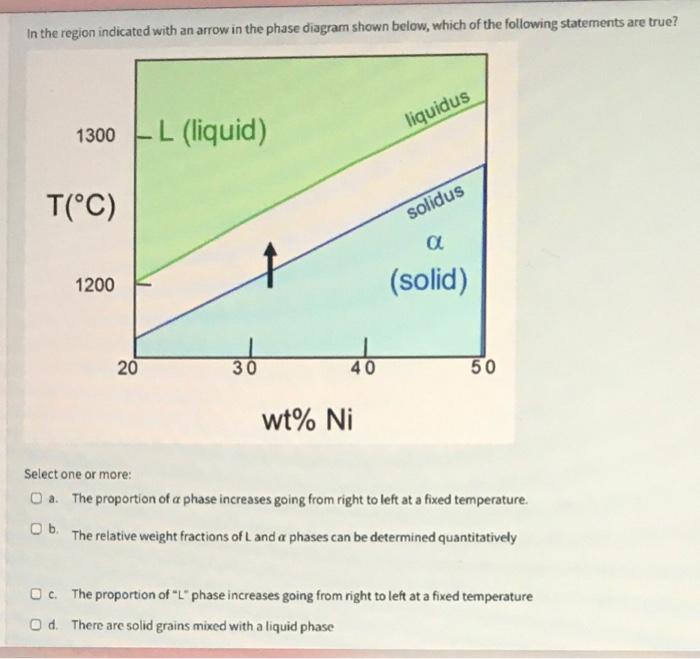
In the region indicated with an arrow in the phase diagram shown below, which of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
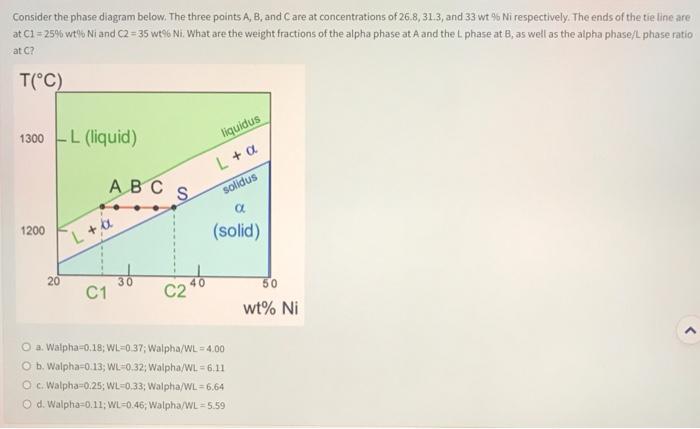
In the region indicated with an arrow in the phase diagram shown below, which of the following statements are true? 1300 L (liquid) - . T(°C) 1200 20 36 40 wt% Ni liquidus solidus α (solid) 50 Select one or more: a. The proportion of a phase increases going from right to left at a fixed temperature. b. The relative weight fractions of L and a phases can be determined quantitatively c. The proportion of "L" phase increases going from right to left at a fixed temperature Od. There are solid grains mixed with a liquid phase Consider the phase diagram below. The three points A, B, and C are at concentrations of 26.8, 31.3, and 33 wt% Ni respectively. The ends of the the line are at C1 = 25% wt% Ni and C2 = 35 wt% Ni. What are the weight fractions of the alpha phase at A and the L phase at B, as well as the alpha phase/L phase ratio at C? T(°C) 1300-L (liquid) 1200 20 ABC L+a 30 C1 liquidus L + a solidus a (solid) S C2 O a. Walpha 0.18; WL-0.37; Walpha/WL=4.00 O b. Walpha-0.13; WL 0.32; Walpha/WL=6.11. O c. Walpha 0.25; WL-0.33; Walpha/WL = 6.64 O d. Walpha=0.11; WL-0.46, Walpha/WL-5.59 50 wt% Ni In the region indicated with an arrow in the phase diagram shown below, which of the following statements are true? 1300 L (liquid) - . T(°C) 1200 20 36 40 wt% Ni liquidus solidus α (solid) 50 Select one or more: a. The proportion of a phase increases going from right to left at a fixed temperature. b. The relative weight fractions of L and a phases can be determined quantitatively c. The proportion of "L" phase increases going from right to left at a fixed temperature Od. There are solid grains mixed with a liquid phase Consider the phase diagram below. The three points A, B, and C are at concentrations of 26.8, 31.3, and 33 wt% Ni respectively. The ends of the the line are at C1 = 25% wt% Ni and C2 = 35 wt% Ni. What are the weight fractions of the alpha phase at A and the L phase at B, as well as the alpha phase/L phase ratio at C? T(°C) 1300-L (liquid) 1200 20 ABC L+a 30 C1 liquidus L + a solidus a (solid) S C2 O a. Walpha 0.18; WL-0.37; Walpha/WL=4.00 O b. Walpha-0.13; WL 0.32; Walpha/WL=6.11. O c. Walpha 0.25; WL-0.33; Walpha/WL = 6.64 O d. Walpha=0.11; WL-0.46, Walpha/WL-5.59 50 wt% Ni
Expert Answer:
Answer rating: 100% (QA)
The given phase diagram having separation for liquid and solid The correct statem... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements are true regarding dividends? _____ 1. Cash and stock dividends reduce retained earnings. _____ 2. Dividends payable is recorded at the time a cash dividend is...
-
Which of the following statements are true of long-term investments? a. They can include investments in trading securities. b. They are always easily sold and therefore qualify as being marketable....
-
Which of the following statements are true for discrete random variables? Explain why each statement is true or false. a. The mean can be negative. b. The mean can be zero. c. The standard deviation...
-
A business student has $3,500 available from a summer job and has identified three potential stocks in which to invest. The cost per share and expected return over the next two years are given in the...
-
A portfolio of stocks generates a 2 9% return in 2008, a 23% return in 2009, and a 17% return in 2010. What is the annualized return (geometric mean) for the entire period?
-
Consider the competition model defined by dx/dt = x(2 - 0.4x - 0.3y) dy/dt = y(1 - 0.1y - 0.3x), where the populations x(t) and y(t) are measured in thousands and t in years. Use a numerical solver...
-
Under CCP 581(b)(1), barring certain exceptions, without a court order a plaintiff has an absolute right to dismiss his or her complaint any time before the commencement of trial. Further investigate...
-
Suppose Nikes managers were considering expanding into producing sports beverages. Why might the company decide to do this under the Nike brand name?
-
What are the implications of shift work, irregular schedules, and long working hours on employee circadian rhythms, sleep quality, and overall health, and what organizational policies and practices...
-
Robson Electronics completed these selected transactions during March 2018: a. Sales of $2,050,000 are subject to an accrued warranty cost of 9%. The accrued warranty payable at the beginning of the...
-
Micromanagement-free zone: DevFacto cofounders Chris Izquerdo and David Cronin aimed to create a company that values open communication and innovation. To do so meant embracing diversity, enticing...
-
Define the concatenation operator of finite displacements. Illustrate with an example.?
-
Provide answers for the following: a ) Explain why a spread spectrum Wi - Fi signal does not create interference with IR networks. b ) What creates interference with IR instead?
-
What is the required shaft power (kW) that must be supplied to pump water at flow rate 0.010 m/s through pipe (fittings are all threaded) with diameter 50 mm? Total length of pipes is 100 m. The pump...
-
Mention one ratio that measures bank profitability and one ratio that measures bank risk. How do you think the two ratios listed above have been influenced by the financial and economic pressures of...
-
Suggest at least 10 business research titles that are easy to conduct and relevant to the COVID-19 pandemic
-
9 mole 9 A=97.80- G=131.29- mole 9 mole 9 mole D=141.98- X = 36.51- 9 mole E 355.68- Z= 35.04-9 mole A canister of gas contains 29.92 grams of gas A, 6.99 grams of gas D and 90.46 grams of gas G. The...
-
The slopes of the tangents at the points where the curve y = x2 - 4x intersects the X-axis is 1) 1 2) +2 3) +3 4) +4
-
Characterize each of the following atoms as being either nucleophilic or electrophilic. (a) Iodide ion, I- (b) Hydrogen ion, H+ (c) Carbon in methyl cation, +CH3 (d) Sulfur in hydrogen sulfide. H2S...
-
The structure of the terpene limonene is shown in the margin. Identify the two 2-methyl-1,3-butadiene (isoprene) units in limonene. (a) Treatment of isoprene with catalytic amounts of acid leads to a...
-
When 1-pentene is treated with mercuric acetate, followed by sodium borohydride, which of the following compounds is the resulting product? (a) 1-Pentyne; (b) pentane; (c) 1-pentanol; (d) 2-pentanol.
-
Assume that Boardmaster sold skateboards to a department store for \($35,000\) cash. How would this transaction affect Boardmasters accounting equation? a. Increase both assets and stockholders...
-
Generally Accepted Accounting Principles (GAAP) are created by the a. Securities and Exchange Commission (SEC). b. Financial Accounting Standards Board (FASB). C. Institute of Management Accountants...
-
Boardmaster is famous for custom skateboards. At the end of a recent year, Boardmasters total assets added up to \($622\) million, and stockholders equity was \($487\) million. How much did...

Study smarter with the SolutionInn App


