12.)Find the molar solubility of BaCro. (K 2.1x10) in a. pure water. b. 25-10 M Na,Cro....
Fantastic news! We've Found the answer you've been seeking!
Question:
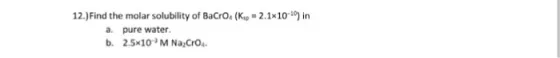
Transcribed Image Text:
12.)Find the molar solubility of BaCro. (K 2.1x10) in a. pure water. b. 25-10 M Na,Cro. 12.)Find the molar solubility of BaCro. (K 2.1x10) in a. pure water. b. 25-10 M Na,Cro.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
The molar solubility of MnCO3 is 4.2 3 1026 M. What is Ksp for this compound?
-
Suppose the molar solubility of nickel hydroxide, Ni(OH)2, is x M. Show that Ksp for nickel hydroxide equals 4x3.
-
a. If the molar solubility of cobalt(II) hydroxide is 5.4 106 mol/L in pure water, what is its Ksp value?
-
The compound bow in the previous problem requires 152 J of work to draw the string back to x = 0.540 m, at which point the archer need only exert 250 N of force to hold the string in place. (a)...
-
Indicate whether the following items would appear on the income statement (IS), statement of owners equity (OE), or balance sheet (BS). a. ______ Tutoring Fees Earned b. ______ Office Equipment c....
-
Prove that the expected number of iterations of the acceptance/rejection method until the first acceptance is k. (Think of each iteration as a Bernoulli trial. What is the expected number of trials...
-
You are appointed to participate on a time efficiency team. The team includes factory workers, Supervisor and a representative from the accounting department. Currently the factory workers record...
-
Suppose selected comparative statement data for the giant bookseller Barnes & Noble are presented here. All balance sheet data are as of the end of the fiscal year (in millions). Instructions Compute...
-
Sam Enterprise provides the following information about its single product, for each month: Estimated Sales Units 3,500 units Selling price per unit $150 Variable cost per unit $90 Total fixed...
-
For the data from Speciatronics, Ltd. (Prob04-09 in Ch04Data.xlsx on the Student Companion Site), the upper specification limit was USL = 5.25, and the lower specification limit was LSL = 1.75....
-
Explain the Planning process group elements: Risk and Procurement Management -Communication and Stakeholde! Management
-
Minnie wants to buy a home. She contacts the owner of a potential property that she likes. While Minnie is not sure that the home is for her, she wants to prevent someone else from buying the...
-
As employees health changes, they may miss work due to illness or injury increasing healthcare cost for employees and employers. Furthermore, employees may be less productive while at work due to...
-
Camembert's commercial paper is currently selling at a discount. It sells for 98.36 of par and matures in 85 days. a. Calculate its yield as quoted in the market. b. Calculate its effective annual...
-
Your mission is to prove / sell the idea of ERP/EIS implementation to the top management & CEO of one of leaders of their industry by describing minimum of below points. By this way you will prove...
-
A company produces industrial rotors and provides the following additional information about steel, which is a direct material for the rotors: Pounds of steel needed per rotor 12 pounds Steel cost...
-
Part (a): Create a discrete probability distribution using the generated data from the following simulator: Anderson, D. Bag of M&M simulator . New Jersey Factory. Click on the simulator to scramble...
-
10m solution. If Ka(HA) = 10 then pOH of solution will be [Given : log4=0.6] (A) 6.7 (B) Greater than 6.7 & less than 7.0 (C) Greater 7.0 & less than 7.3 (D) Greater than 7.3
-
Since 1998, elements 112, 114, and 116 have been synthesized. Element 112 was created by bombarding 208Pb with 66Zn; element 114 was created by bombarding 244Pu with 48Ca; element 116 was created by...
-
State whether each of the following properties of the representative elements generally increases or decreases (a) From left to right across a period and (b) From top to bottom within a group:...
-
What is the coordination number of each sphere in (a) A simple cubic cell, (b) A body-centered cubic cell, (c) A face-centered cubic cell? Assume the spheres are all the same?
-
Hardwoods, Inc. purchased a tract of timber for a total cost of \($500,000\). The tract is estimated to contain 200,000 board feet of lumber once the timber is harvested. Hardwoods expects the land...
-
Match the descriptive explanation below with the correct term: 1. An exclusive right that protects an owner against the unauthorized reproduction of a specific written work. 2. The periodic write-off...
-
Rollins Co. reported the following information in its 2018 financial statements: Compute the return on assets and asset turnover for Rollins Co. in 2018. Net sales. Net income Beginning of year total...

Study smarter with the SolutionInn App



