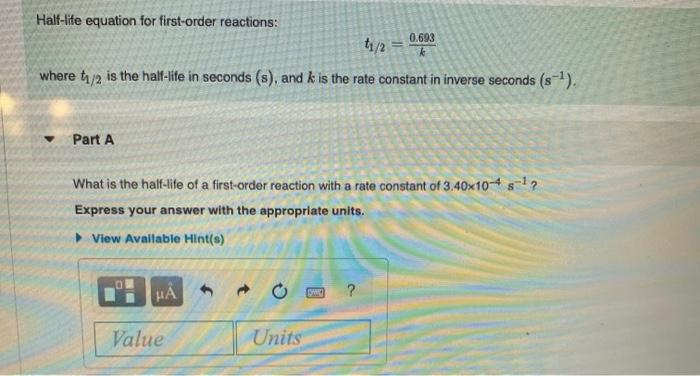
Half-life equation for first-order reactions: t/2 = where t/2 is the half-life in seconds (s), and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
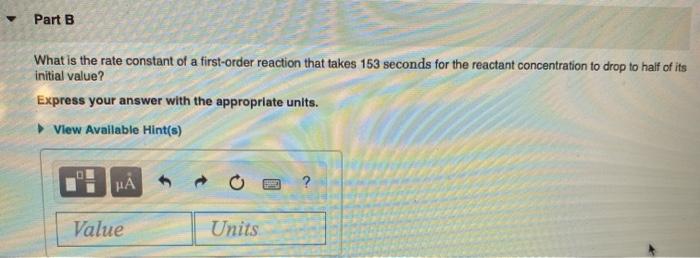
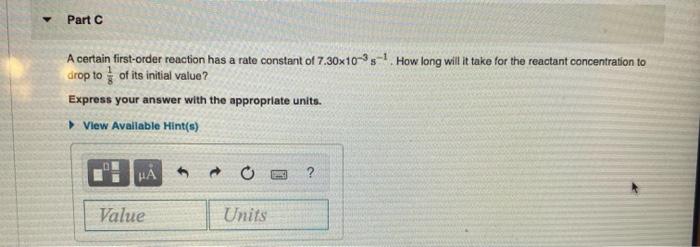
Half-life equation for first-order reactions: t₁/2 = where t₁/2 is the half-life in seconds (s), and k is the rate constant in inverse seconds (s-¹). Part A What is the half-life of a first-order reaction with a rate constant of 3.40x104s¹? Express your answer with the appropriate units. View Available Hint(s) HÅ Value Units 0.693 (PHIC) ? Part B What is the rate constant of a first-order reaction that takes 153 seconds for the reactant concentration to drop to half of its initial value? Express your answer with the appropriate units. View Available Hint(s) μà Value Units Part C A certain first-order reaction has a rate constant of 7.30x10-3s¹ How long will it take for the reactant concentration to drop to of its initial value? Express your answer with the appropriate units. ▸ View Available Hint(s) HA Value 0 Units Half-life equation for first-order reactions: t₁/2 = where t₁/2 is the half-life in seconds (s), and k is the rate constant in inverse seconds (s-¹). Part A What is the half-life of a first-order reaction with a rate constant of 3.40x104s¹? Express your answer with the appropriate units. View Available Hint(s) HÅ Value Units 0.693 (PHIC) ? Part B What is the rate constant of a first-order reaction that takes 153 seconds for the reactant concentration to drop to half of its initial value? Express your answer with the appropriate units. View Available Hint(s) μà Value Units Part C A certain first-order reaction has a rate constant of 7.30x10-3s¹ How long will it take for the reactant concentration to drop to of its initial value? Express your answer with the appropriate units. ▸ View Available Hint(s) HA Value 0 Units
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below ... View the full answer

Related Book For 

Discovering Advanced Algebra An Investigative Approach
ISBN: 978-1559539845
1st edition
Authors: Jerald Murdock, Ellen Kamischke, Eric Kamischke
Posted Date:
Students also viewed these chemistry questions
-
How long will it take for monthly payments interest rate on the loan is: a. 6% compounded monthly? b. 7% compounded monthly? c. 8% compounded monthly? d. 9% compounded monthly
-
How long will it take for monthly payments interest rate on the loan is: a. 7.5% compounded annually? b. 7.5% compounded semiannually? c. 7.5% compounded quarterly? d. 7.5% compounded monthly?
-
How long will it take a 1750-W motor to lift a 315-kg piano to a sixth-story window 16.0m above?
-
A Moving to another question will save this response. Question 9 1 L The absent of the logic action Release in the Resource Module means: The operator will rework the entity a. b. C. The operator...
-
How can too little group cohesiveness result in low levels of performance?
-
Add 10 to each of the scores in Question 4.4 (1, 3, 4, 4) to produce a new distribution (11, 13, 14, 14). Would you expect the value of the sample standard deviation to be the same for both the...
-
In January 1984, Richard Goose Gossage signed a contract to play for the San Diego Padres that guaranteed him a minimum of \($9,955,000.\) The guaranteed payments were \($875,000\) for 1984,...
-
Upton Computers makes bulk purchases of small computers, stocks them in conveniently located warehouses, and ships them to its chain of retail stores. Upton's balance sheet as of December 31, 2004,...
-
Future Value: The State of Confusion wants to change the currentretirement policy for state employees. To do? so, however, thestate must pay the current pension fund members the present valueof t 2...
-
1. Discuss the main issues faced by Yalla Momos. 2. Evaluate the current financial performance of Yalla Momos and compare his performance to the industry ratios. 3. Elaborate on the relevant factors...
-
Weston Products manufactures an industrial cleaning compound that goes through three processing departmentsGrinding, Mixing, and Cooking. All raw materials are introduced at the start of work in the...
-
A bar is subjected to the following stresses \[ \sigma_{x}=50 \mathrm{MPa}, \sigma_{y}=-40 \mathrm{MPa}, \sigma_{z}=70 \mathrm{MPa} \] If \(v=0.25\) and \(E=200 \mathrm{GPa}\), calculate (a) the...
-
State parallel axis theorem.
-
What is moment of resistance ?
-
A square bar of \(20 \mathrm{~mm}\) side and \(200 \mathrm{~mm}\) long is subjected to a compressive load of 200 \(\mathrm{KN}\) applied in the direction of its length. If all the strains in the...
-
Define bending stiffness
-
MODULE 4 Activity #1 Discussion Sheet Video Discussion Sheet Video Source: https://www.youtube.com/watch?v=leLF0FnHFUk What are some of the symptoms of the illness that are described? How did the...
-
What are three disadvantages of using the direct write-off method?
-
Decide whether each expression is an identity by substituting values for A and B. a. cos (A + B) = cos A + cos B b. sin (A + B) = sin A + sin B c. cos (2A) = 2 cos A d. sin (2A) = 2 sin A
-
Create a data set of 9 values such that the median is 28, the minimum is 11, and there is no upper whisker on a box plot of the data.
-
Los Angeles, California, and Honolulu, Hawaii, are about 2500 mi apart. One plane flies from Los Angeles to Honolulu, and a second plane flies in the opposite direction. a. Describe the meaning of...
-
Take each figure in the 2001 columns and measure its percentage change over 2000. Identify where the most signifi- cant changes have occurred.
-
As with the balance sheet, compare the current-year reported figures in the profit and loss account with the information shown for the prior year. Calculate the percentage change from one year to the...
-
What two events led to the transformation and spread of accounting between the 12th and 15th centuries?

Study smarter with the SolutionInn App


