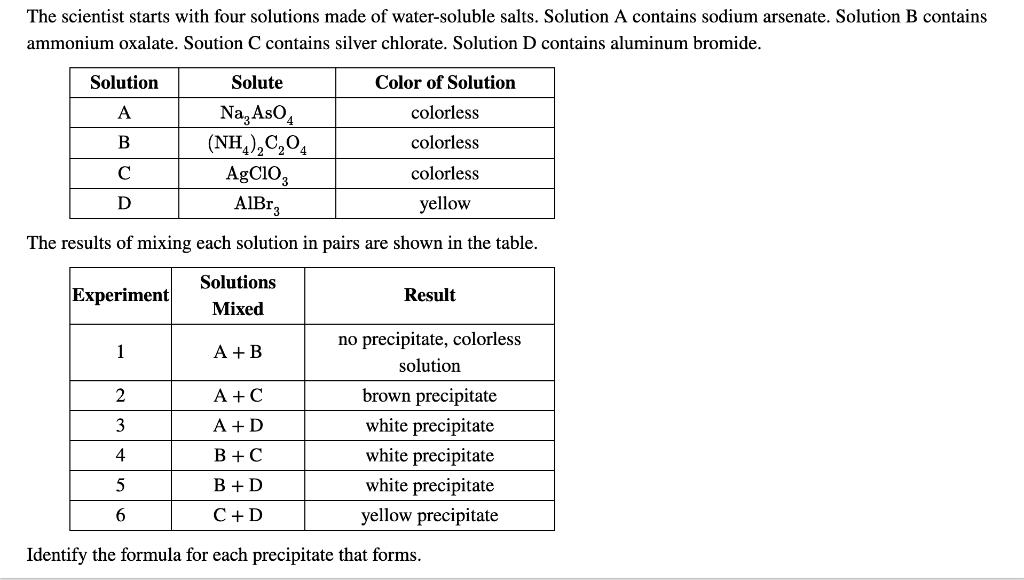
The scientist starts with four solutions made of water-soluble salts. Solution A contains sodium arsenate. Solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
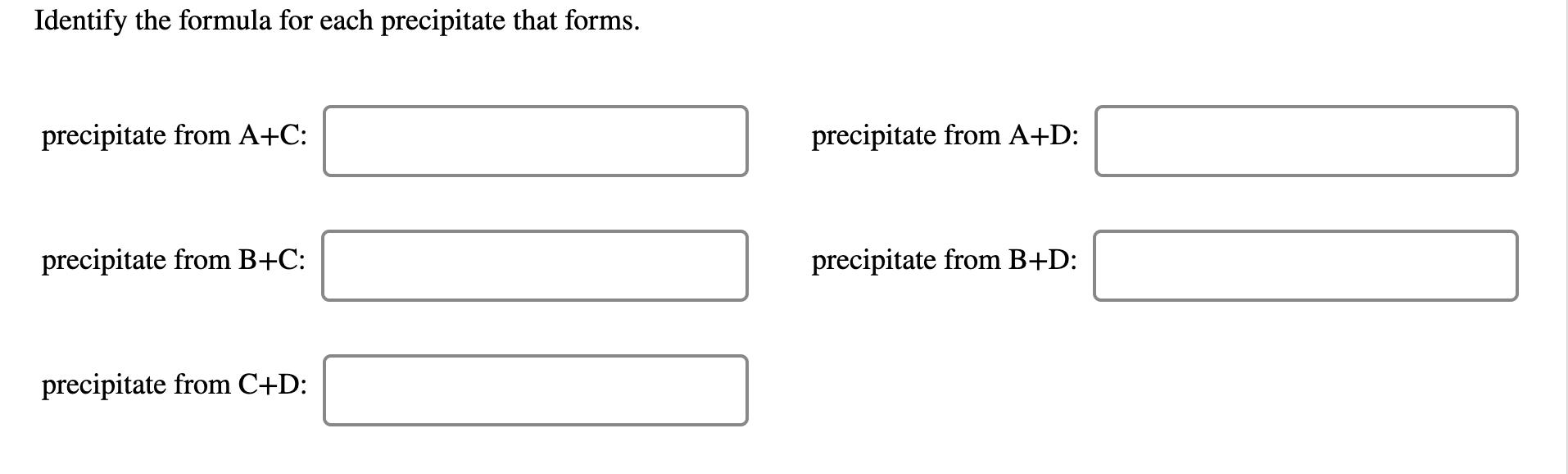
The scientist starts with four solutions made of water-soluble salts. Solution A contains sodium arsenate. Solution B contains ammonium oxalate. Soution C contains silver chlorate. Solution D contains aluminum bromide. Solution A B с D Solute Color of Solution Na, AsO4 colorless (NH4)2C₂O4 colorless AgClO3 colorless AlBrą yellow The results of mixing each solution in pairs are shown in the table. Experiment 1 2 3 4 6 Solutions Mixed A + B A+C A+D B+C B+D C + D Result no precipitate, colorless solution brown precipitate white precipitate white precipitate white precipitate yellow precipitate Identify the formula for each precipitate that forms. Identify the formula for each precipitate that forms. precipitate from A+C: precipitate from B+C: precipitate from C+D: precipitate from A+D: precipitate from B+D: The scientist starts with four solutions made of water-soluble salts. Solution A contains sodium arsenate. Solution B contains ammonium oxalate. Soution C contains silver chlorate. Solution D contains aluminum bromide. Solution A B с D Solute Color of Solution Na, AsO4 colorless (NH4)2C₂O4 colorless AgClO3 colorless AlBrą yellow The results of mixing each solution in pairs are shown in the table. Experiment 1 2 3 4 6 Solutions Mixed A + B A+C A+D B+C B+D C + D Result no precipitate, colorless solution brown precipitate white precipitate white precipitate white precipitate yellow precipitate Identify the formula for each precipitate that forms. Identify the formula for each precipitate that forms. precipitate from A+C: precipitate from B+C: precipitate from C+D: precipitate from A+D: precipitate from B+D:
Expert Answer:
Answer rating: 100% (QA)
Answer AC Na3AsO4 3 AgCIO3 NaClO3 aq here the precipitate is Ag3AsO4 BC Ag3As... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Evertight a leading manufacturer of quality nails produces 1 2 3 4 and 5 inch nails clavos for various uses In the production process if there is an overrun or the nails are slightly defective they...
-
Consider the following time series: t 1 2 3 4 5 6 7 yt 120 110 100 96 94 92 88 A. Construct a time series plot. What type of pattern exists in the data? B. Use excel solver or lingo to find the...
-
Given the following data: Period 1 2 3 4 5 6 7 8 9 Gross Requirements 70 30 90 30 80 20 90 60 80 The beginning inventory is 0 and there are no scheduled receipts. The order cost is $90 and the...
-
Write the complete APT part program to profile mill the outside edges of the part. The part is 15 mm thick. Tooling = 30 mm diameter end mill with four teeth, cutting speed = 150 mm/min, and feed =...
-
Sams Cat Hotel operates 52 weeks per year, 6 days per week, and uses a continuous review inventory system. It purchases kitty litter for $11.70 per bag. The following information is available about...
-
According to some economists, why might business firms pay wage rates above market-clearing levels?
-
Onstar Communication issued \(\$ 100,000\) of \(6 \%, 10\)-year bonds payable on October 1, 2008, at par value. Onstar's accounting year ends on December 31. Journalize the following transactions....
-
Calculate the annual cash dividends required to be paid for each of the following preferred stock issues: Required: a. $3.75 cumulative preferred, no par value; 200,000 shares authorized, 161,522...
-
Quarantine Company manufactures Part AA for use in its production cycle. The costs per unit for 25,000 units for the part are as follows: Direct materials P 7.50 Direct labor Variable overhead 37.50...
-
Computer Service and Repair was started five years ago by two college roommates. The company's comparative balance sheets and income statement are presented below, along with additional information....
-
Rob, convinced of his wife Pauline's infidelity, decides to kill three of her supposed lovers, Xavier, Yusuf and Zane. Rob persuades Xavier to take a train to London because he has hired Hitman, a...
-
Although the validity of evidential matter is dependent on the circumstances under which it is obtained, there are three general presumptions that have some usefulness. The situations given below...
-
Describe the principal stages of a general-purpose operational amplifier.
-
In each of the following cases involving non statistical sampling, indicate what conclusions the auditor might draw and why. a. The likely misstatement is more than the tolerable misstatement. b. The...
-
Which of the following statements is correct concerning PPS sampling? a. The sampling distribution should approximate the normal distribution. b. Overstated units have a lower probability of sample...
-
What are the differences between object-oriented modeling and using data flow diagrams?
-
Crane Services Corporation had the following accounts and balances: Accounts payable Accounts receivable Buildings Cash $29000 4800 Land 69800 Equipment 15150 Unearned service revenue Total...
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
When magnesium metal is burned in air (Figure 3.6), two products are produced. One is magnesium oxide, MgO. The other is the product of the reaction of Mg with molecular nitrogen, magnesium nitride....
-
Propylene, C3H6, is a gas that is used to form the important polymer called polypropylene. Its Lewis structure is (a) What is the total number of valence electrons in the propylene molecule? (b) How...
-
Which substances in Table 10.3 would you expect to deviate most from ideal-gas behavior at low temperature and high pressure? Which would deviate least? Explain. TABLE 10.3Van der Waals Constants for...
-
Estimate the gravitational force and the acceleration due to gravity on a body of \(1.25 \mathrm{~kg}\) mass on the earth's surface. The radius and mass of the earth are \(6370 \mathrm{~km}\) and...
-
A reactor contains a gas mixture of \(25 \mathrm{~kg} \mathrm{NH}_{3}, 15 \mathrm{~kg} \mathrm{CO}\) and \(10 \mathrm{~kg} \mathrm{C}_{2} \mathrm{H}_{2}\). Calculate the total number of moles of the...
-
Estimate the gravitational force on a body of \(1.5 \mathrm{~kg}\) mass on the earth's surface, given that the radius and mass of the earth are \(6000 \mathrm{~km}\) and \(6 \times 10^{24}...

Study smarter with the SolutionInn App


