Help me to do question 5. Thank you. The company that we will work on in FEDEX.
Fantastic news! We've Found the answer you've been seeking!
Question:
Help me to do question 5. Thank you.
Transcribed Image Text:
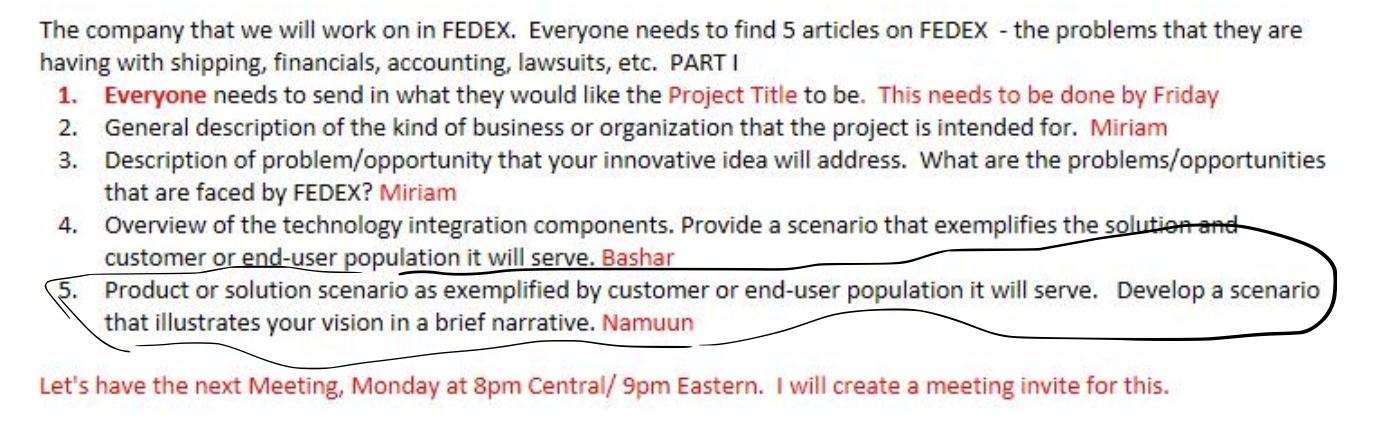
The company that we will work on in FEDEX. Everyone needs to find 5 articles on FEDEX - the problems that they are having with shipping, financials, accounting, lawsuits, etc. PARTI 1. Everyone needs to send in what they would like the Project Title to be. This needs to be done by Friday 2. General description of the kind of business or organization that the project is intended for. Miriam 3. Description of problem/opportunity that your innovative idea will address. What are the problems/opportunities that are faced by FEDEX? Miriam 4. Overview of the technology integration components. Provide a scenario that exemplifies the solution and customer or end-user population it will serve. Bashar Product or solution scenario as exemplified by customer or end-user population it will serve. Develop a scenario that illustrates your vision in a brief narrative. Namuun Let's have the next Meeting, Monday at 8pm Central/ 9pm Eastern. I will create a meeting invite for this. The company that we will work on in FEDEX. Everyone needs to find 5 articles on FEDEX - the problems that they are having with shipping, financials, accounting, lawsuits, etc. PARTI 1. Everyone needs to send in what they would like the Project Title to be. This needs to be done by Friday 2. General description of the kind of business or organization that the project is intended for. Miriam 3. Description of problem/opportunity that your innovative idea will address. What are the problems/opportunities that are faced by FEDEX? Miriam 4. Overview of the technology integration components. Provide a scenario that exemplifies the solution and customer or end-user population it will serve. Bashar Product or solution scenario as exemplified by customer or end-user population it will serve. Develop a scenario that illustrates your vision in a brief narrative. Namuun Let's have the next Meeting, Monday at 8pm Central/ 9pm Eastern. I will create a meeting invite for this.
Expert Answer:
Answer rating: 100% (QA)
Answer It looks like youre organizing a project related to FedEx and its various aspects including p... View the full answer

Related Book For 

Posted Date:
Students also viewed these marketing questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
The United StatesMexicoCanada Agreement replaced what trade agreement?
-
Tastyfreeze Company is a small producer of fruitflavored frozen desserts. For many years, its products have had strong regional sales because of brand recognition; however, other companies have begun...
-
Prior to the 2010 Dodd-Frank Act "Volker Rule", banks employed proprietary traders (aka Prop Traders) whose job was to invest the Bank's capital in various short-term trading strategies. Prop Traders...
-
Limo.com had the following balances on December 31, 2010, before the year- end adjustments: Accounts Receivable 104,000 Allowance for Uncollectible Accounts 1,300 The aging of receivables yields...
-
Refer to Exercise 3-27A. Requirements 1. Use the data in the partial worksheet to prepare Winwood Production Company's classified balance sheet at December 31 of the current year. Use the report...
-
Required information [The following information applies to the questions displayed below.] Lexon Inc. is a large manufacturer of affordable DVD players. Management recently became aware of rising...
-
What is the key difference between the change in management from banks in relation to their sources of funds before 1980s and after 1980s. O Instead of managing their own funding to meet loan demand,...
-
Write the following as a single rational expression. 1 1 X X-2
-
Consider these molecules: CHA, NCl3, BF3, CS2 (a) In which compound are the bonds most polar? (b) Which compounds in the list are not polar? (c). Explain in terms of noncovalent interactions why...
-
Xavier and Yolanda have original investments of $51,200 and $103,600, respectively, in a partnership. The articles of partnership include the following provisions regarding the division of net...
-
QUESTION 1 Given the following information: Units produced Materials (kg) Material Costs Direct Labour (Hours) Original Budget 1,000 units 400 kg $8,000 Actual 1,070 units 409 kg See other info...
-
Why would Qantas international (not domestic) have a underlying earnings before interest and tax (EBIT) of $-238,000,000
-
In Caba Beach Resort, a boat ride cost P^(300) intended for 10 passengers only and additional passengers are charged P^(30) per head. a. Find the fare function f(x) where x represents the number of...
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
A group of investors in your city is considering opening a new upscale supermarket to compete with the major supermarket chains that are currently dominating the citys marketplace. They have called...
-
You supervise a team of marketing analysts who work on different snack products in a large food products company. The marketing analysts have recently received undergraduate degrees in business or...
-
Youre the manager of sales in an expensive sports car dealership. A young executive who has just received a promotion comes in and wants to buy a car that you know is out of her price range. Do you...
-
Show that \[ \gamma_{i}=\frac{\bar{\phi}_{i}}{\phi_{i}} \]
-
Carbon dioxide occupies a tank at \(100^{\circ} \mathrm{C}\). If the volume of the tank is \(0.5 \mathrm{~m}^{3}\) and the pressure is \(500 \mathrm{kPa}\), determine the mass of the gas in the tank.
-
If a cylinder of volume \(0.1 \mathrm{~m}^{3}\) is filled with \(1.373 \mathrm{~kg}\) of ammonia at \(1.95 \mathrm{MPa}\), determine the temperature at which ammonia exists in the cylinder. Assume...

Study smarter with the SolutionInn App


