Help on these two 8. At 500.0 K, one mole of gaseous ONCI is placed in a
Fantastic news! We've Found the answer you've been seeking!
Question:
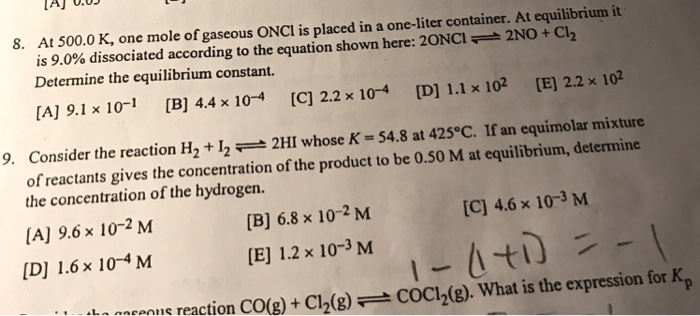
Help on these two
Transcribed Image Text:
8. At 500.0 K, one mole of gaseous ONCI is placed in a one-liter container. At equilibrium it is 9.0% dissociated according to the equation shown here: 20NCI 2NO + Cl₂ Determine the equilibrium constant. [A] 9.1 x 10-1 [B] 4.4 x 10-4 [C] 2.2 x 10-4 [D] 1.1 x 10² [E] 2.2 x 102 9. Consider the reaction H₂ + 122HI whose K = 54.8 at 425°C. If an equimolar mixture of reactants gives the concentration of the product to be 0.50 M at equilibrium, determine the concentration of the hydrogen. [A] 9.6 × 10-2 M [D] 1.6 x 10-4 M [C] 4.6 x 10-3 M 1-(1+1) = -1 ons reaction CO(g) + Cl₂(g) COC1₂(g). What is the expression for Kp [B] 6.8 x 10-2 M [E] 1.2 x 10-3 M 8. At 500.0 K, one mole of gaseous ONCI is placed in a one-liter container. At equilibrium it is 9.0% dissociated according to the equation shown here: 20NCI 2NO + Cl₂ Determine the equilibrium constant. [A] 9.1 x 10-1 [B] 4.4 x 10-4 [C] 2.2 x 10-4 [D] 1.1 x 10² [E] 2.2 x 102 9. Consider the reaction H₂ + 122HI whose K = 54.8 at 425°C. If an equimolar mixture of reactants gives the concentration of the product to be 0.50 M at equilibrium, determine the concentration of the hydrogen. [A] 9.6 × 10-2 M [D] 1.6 x 10-4 M [C] 4.6 x 10-3 M 1-(1+1) = -1 ons reaction CO(g) + Cl₂(g) COC1₂(g). What is the expression for Kp [B] 6.8 x 10-2 M [E] 1.2 x 10-3 M
Expert Answer:
Answer rating: 100% (QA)
8 20 HC Intial Im 1X H I 9 2NOC1 at equilbirium 091 9x g... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
These two pie charts show the origin of cars bought by customers who traded in domestic and Asian models. For example, among those trading in a car produced by a domestic manufacturer, 29% purchased...
-
Solve these two Differential Equations?? 1. y' = 2. y' = +3y 2xy x 4x + 3y 2x + y
-
Help & Save is a private not-for-profit organization that operates in Kansas. Swim For Safety is a private not-for-profit organization that operates in Missouri. The leaders of these two...
-
Assume a retailer has fixed costs of $10,000, a unitvariable cost of $25, and a 50% retail margin. How many units must be sold for her tobreak-even? If she has a target profit of $200,000, how many...
-
1. Which Big Five personality traits best describe Daphne? Give examples of behavior from the film scene to support your observations. 2. Which Big Five personality traits best describe Millie? Give...
-
Locate the following tax services in your library, and indicate the name of the publisher: a. United States Tax Reporter. b. Standard Federal Tax Reporter. c. Federal Tax Coordinator 2d. d. Mertens...
-
Simplify the irrational number \(\sqrt{330}\) and express in lowest terms. Identify the rational and irrational parts.
-
Recording Seven Typical Adjusting Entries Dittmans Variety Store is completing the accounting process for the year just ended, December 31, 2011. The transactions during 2011 have been journalized...
-
You identify a EuroRail bond that you want to invest in. You'd like to buy $1,000 par value. The bond matures in 10 years, has a coupon of 4%, and pays interest semiannually. If you pay $934 for th...
-
= On 1/1/X4, Phillip invested $1,100,000 in Sleeper's ordinary shares (35% owned). Sleeper reported: Assets $3,500,000 Liabilities 600,000 The book value of Sleeper's net assets approximated market...
-
When evaluating costs as to whether your company should build "in house" or contract it externally, what should the Objective Function look like? O Profit/Loss = PurchaseTotal...
-
SELECT * FROM CUSTOMERS JOIN ORDERS ON CUSTOMERS.customer_id = ORDERS.customer_id JOIN PRODUCTS ON ORDERS.product_id = PRODUCTS.product_id; IT SAYS NO DATA FOUND
-
simplify - 4 2 + 3 - 1 6 2
-
Describe the distinct mechanisms by which organelles such as the mitochondria and the endoplasmic reticulum are formed, and discuss how these processes are regulated during cell division .
-
The financial manager of GCC Cereals is in the process of preparing a cash budget for the first quarter of 2021. The firm typically sells 1/3 of its monthly sales on cash terms and the rest on...
-
Find the volume of the solid generated by rotating the region bounded by the curve f (x)=cos and the lines Type your answer... = and the x-axis, around the x-axis Find the volume of the solid...
-
Read the Case Study for "Johnny" and review/score the most current version of DSM Parent/Guardian-Rated Level 1 Cross-Cutting Symptom MeasureChild Age 6-17 form completed by the mother (attached)....
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
Photogray lenses incorporate small amounts of silver chloride in the glass of the lens. When light hits the AgCl particles, the following reaction occurs: The silver metal formed causes the lenses to...
-
The memory metal, nitinol, is an alloy of nickel and titanium. It is called a memory metal because after being deformed, a piece of nitinol wire will return to its original shape. (See Chem. Matters,...
-
Which of the following sets of quantum numbers are not allowed in the hydrogen atom? For the sets of quantum numbers that are incorrect, state what is wrong in each set. a. n = 3, = 2, m = 2 b. n =...
-
Using Google, determine the number of hits for these terms: a. Forensic accounting. b. Fraud auditing. c. Forensic auditing. d. Fraud examination.
-
Search the Internet: Is plagiarism fraud?
-
Search the Internet and learn about the AICPA's site on Business Valuation and Forensic/Litigation Services.

Study smarter with the SolutionInn App


