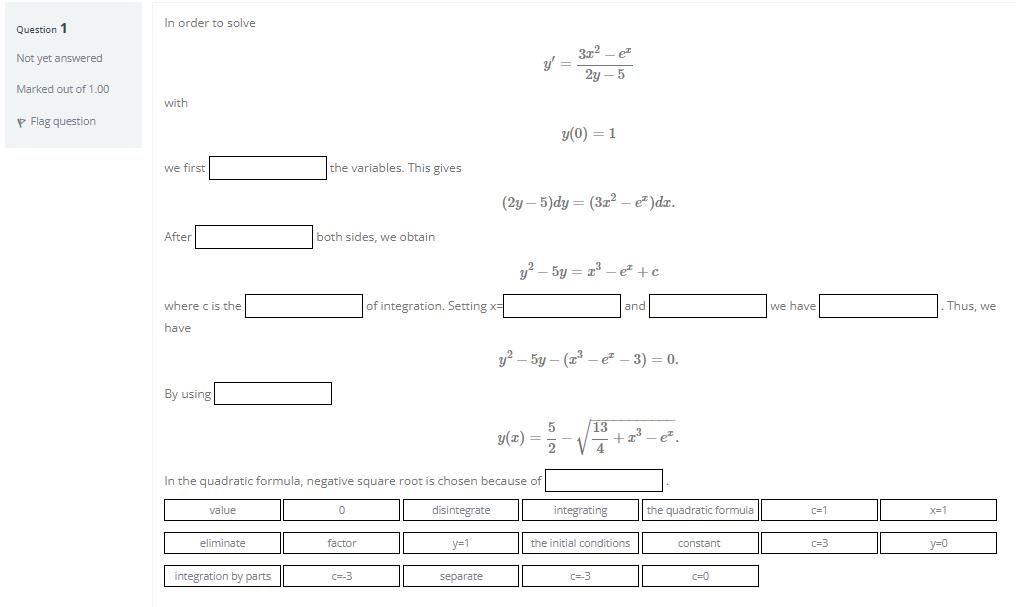
Question 1 Not yet answered Marked out of 1.00 Flag question In order to solve with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question 1 Not yet answered Marked out of 1.00 Flag question In order to solve with y(0) = 1 we first (2y-5)dy = (3x² e² )dx. After y²-5y = ³e +c of integration. Setting x= and where c is the have y²-5y-(x-e- 3) = 0. By using 13 y(x)= 5 2 +2²³-e². In the quadratic formula, negative square root is chosen because of value 0 disintegrate y=1 eliminate factor integration by parts c=-3 separate the variables. This gives both sides, we obtain y' = 3r² p² 2y-5 integrating the initial conditions c=3 the quadratic formula constant c=0 we have c=1 c=3 Thus, we x=1 y=0 Question 1 Not yet answered Marked out of 1.00 Flag question In order to solve with y(0) = 1 we first (2y-5)dy = (3x² e² )dx. After y²-5y = ³e +c of integration. Setting x= and where c is the have y²-5y-(x-e- 3) = 0. By using 13 y(x)= 5 2 +2²³-e². In the quadratic formula, negative square root is chosen because of value 0 disintegrate y=1 eliminate factor integration by parts c=-3 separate the variables. This gives both sides, we obtain y' = 3r² p² 2y-5 integrating the initial conditions c=3 the quadratic formula constant c=0 we have c=1 c=3 Thus, we x=1 y=0
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
Question 1 Not yet answered Marked out of 1.00 Flag question In order to solve with y(0) = 1 we first (2y-5)dy = (3x e )dx. After y-5y = e +c of integration. Setting x= and where c is the have...
-
In order to solve this problem, you must read the description of the Hammett Ï, Ï treatment given in Chapter 17, Problem 76. When the rate constants for the hydrolysis of several morpholine...
-
Question 7 Not yet answered Marked out of 2.00 Flag question The state-differential equation of the above signal-flow graph is: Select one: ree a. %3B b. d. None of these answers is correct. .e.
-
Watts and Strogatz proposed a hybrid model that contains typical links of vertices near each other (people know their geographic neighbors), plus some random long-range connection links. Plot the...
-
Alliances are often used to pursue business-level goals, but they may be managed at the corporate level. Explain why this portfolio approach to alliance management would make sense.
-
Renee operates a proprietorship selling collectibles over the Web. This year, Renee's business reported revenue of $95.5 million and deducted $88.6 million in expenses and loss carryovers. Her...
-
What authorizations are required for payroll transactions? For each authorization identify the department or individual who should make the authorization.
-
The catering manager of La Vista Hotel, Lisa Ferguson, is disturbed by the amount of silverware she is losing every week. Last Friday night, when her crew tried to set up for a banquet for 500...
-
1- Workpapers (year of adquisicion, fair value/book value differentials, intercompanu balances) Pik Corporation acquired 80 percent of Sel Corporation's common stock on January 1, 2011, for $210,000...
-
Consider the December transactions for Crystal Clear Cleaning that were presented in Chapter 5. Crystal Clear uses the perpetual inventory system. Dec. 2 Purchased 475 units of inventory for $2,850...
-
Calculate the portfolio variance if you have 75% in Security B and 25% in Security C. Year A, % B, % C, % D, % E, % 1 S&P, % 10.67-3.76 12.98 25.51 8.96 0.34 2 12.54 21.67 45.23 18.97 29.67 15.94 3...
-
An individual or fi rm in the business of transporting goods between certain points for anyone. a. bailee b. bailment c. bailor d. carriers lie n e. common c arrier f. consignee g. consignor h....
-
A relationship in which the bailee comes into possession by having goods thrust upon him or her. a. bailee b. bailment c. bailor d. carriers lie n e. common c arrier f. consignee g. consignor h....
-
A transfer of the tenants interest for part of the premises or for part of the term of the lease. a. abandonment b. assignment c. condition d. covenant e. eviction f. lease g. lessee h. lessor i....
-
The Market Potential Index (MPI) is an indexing study conducted by the Michigan State University Center for International Business Education and Research (MSU-CIBER) to compare emerging markets on a...
-
The practice of a court to follow previous decisions. a. administrative law b. appellate court c. civil law d. common law e. criminal law f. defendants g. express powers h. implied powers i. original...
-
The major reason why organizational memory applications do not succeed is: Question 3 options: A) The context of content is neglected, making it difficult for employees to decide whether the...
-
Feller Company purchased a site for a limestone quarry for $100,000 on January 2, 2019. It estimate that the quarry will yield 400,000 tons of limestone. It estimates that its retirement obligation...
-
When phenol is treated with Br2, a mixture of monobromo-, dibromo-, and tribromophenols is obtained. Design a synthesis that would convert phenol primarily to orthobromophenol.
-
Show the mechanism for alkylation of benzene by an alkene.
-
Calculate the amount of steric strain in each of the chair conformers of 1,1,3-trimethylcyclohexane. Which conformer would predominate at equilibrium?
-
The reaction \(A(\mathrm{l}) ightarrow R(\mathrm{~g})\) is allowed to reach equilibrium condition in an autoclave. At equilibrium there are two phases - one a pure liquid phase of \(A\) and the other...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...
-
The internal energy of an ideal gas depends on (a) Temperature, specific heat and volume (b) Temperature and specific heat (c) Temperature, specific heat and pressure (d) Pressure, volume and...

Study smarter with the SolutionInn App



