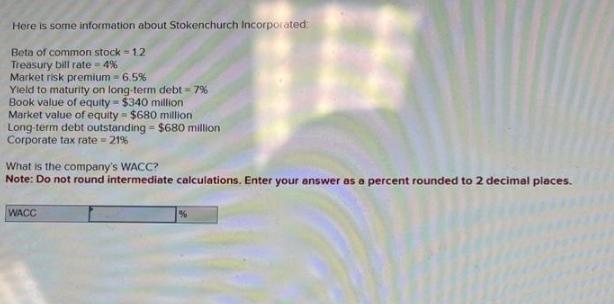
Here is some information about Stokenchurch Incorporated Beta of common stock - 1.2 Treasury bill rate=4%...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Here is some information about Stokenchurch Incorporated Beta of common stock - 1.2 Treasury bill rate=4% Market risk premium - 6.5% Yield to maturity on long-term debt = 7% Book value of equity = $340 million Market value of equity - $680 million Long-term debt outstanding - $680 million Corporate tax rate=21% What is the company's WACC? Note: Do not round intermediate calculations. Enter your answer as a percent rounded to 2 decimal places. WACC Here is some information about Stokenchurch Incorporated Beta of common stock - 1.2 Treasury bill rate=4% Market risk premium - 6.5% Yield to maturity on long-term debt = 7% Book value of equity = $340 million Market value of equity - $680 million Long-term debt outstanding - $680 million Corporate tax rate=21% What is the company's WACC? Note: Do not round intermediate calculations. Enter your answer as a percent rounded to 2 decimal places. WACC
Expert Answer:
Answer rating: 100% (QA)
The companys WACC can be calculated using the following formula WACC Market value ... View the full answer

Related Book For 

Corporate Finance
ISBN: 9781260772388
13th Edition
Authors: Stephen Ross, Randolph Westerfield, Jeffrey Jaffe
Posted Date:
Students also viewed these finance questions
-
Identify and discuss the role of theory in criminological/criminal justice research. Furthermore, discuss why there has been such a shortage of new theories since the 1960s. 2. Select a recent...
-
The treasurer of Kelly Bottling Company (a corporation) currently has $200,000 invested in preferred stock yielding 10 percent. He appreciates the tax advantages of preferred stock and is considering...
-
Describe what this statement does: print user name = + userName
-
The potential energy of the rotation of one CH3 group relative to its neighbour in ethane can be expressed as V( rp)= Vo cos 3rp. Show that for small displacements the motion of the group is harmonic...
-
Why would you want to change the column width and/or row height of a table?
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the annual worth of this investment? b....
-
Horizon Cellular manufactures cell phones for exclusive use in its communication network. Management must select a circuit board supplier for a new phone soon to be introduced to the market. The...
-
Find the perimeter of the figure shown. Express the perimeter using the same unit of measure that appears on the given sides. 13 yd Perimeter = 5 yd 5 yd 13 yd 18 yd 18 yd
-
Fill in the missing amounts from the following T accounts. Accounts Receivable Accounts Payable Sales Aug. 10 17,500 15 6,500 Aug. 5 (c) Aug. 10 50,000 18 3,400 Aug. 12 500 Aug. 23 (a) Aug. 29 5,800...
-
Prepare a broad audit plan: COCA-COLA Co. a. What material types of transactions and transaction cycles are involved? b. What are the high-risk areas? c. What are the low-risk areas? d. If management...
-
In December 2015, Apple had cash of $38.42 billion, current assets of $76.47 billion, and current liabilities of $76.29 billion. It also had inventories of $2.45 billion. a. What was Apples current...
-
Large firms and smaller entrepreneurial firms play different roles in business and society and can often produce the best results by partnering with each other rather than acting as adversaries. The...
-
1 mol of steam is initially at 10 bar and 200C. The surroundings are at 20C and 1 bar. (a) Calculate the exergy of the system. (b) Calculate the change in exergy for a process where the steam is...
-
In December 2015, General Electric (GE) had a book value of equity of $97.6 billion, 9.6 billion shares outstanding, and a market price of $32.79 per share. GE also had cash of $103.4 billion, and...
-
Steam enters a turbine with a mass fl ow rate of 5 [kg/s]. The inlet pressure is 60 bar, and the inlet temperature is 500C. The outlet contains saturated steam at 1 bar. The surroundings are at 20C....
-
Which of the following sets of vectors are lin-early independent? {{:][ } } -3 A. 6. 4 8 B. {[ *):[ ; } {[}[]-[? -9 C. 7 -7 } {[ : }{} B D. -9- 7 | 1 - E. 4 -7 9. . 3 -8 6. 4 -5
-
Suppose the S&P 500 futures price is 1000, = 30%, r = 5%, = 5%, T = 1, and n = 3. a. What are the prices of European calls and puts for K = $1000? Why do you find the prices to be equal? b. What...
-
Gasworks, Inc., has been approached to sell up to 5 million gallons of gasoline in three months at a price of $1.85 per gallon. Gasoline is currently selling on the wholesale market at $1.63 per...
-
Edgehill, Inc., has 355,000 bonds outstanding. The bonds have a par value of $1,000, a coupon rate of 5.4 percent paid semiannually, and 27 years to maturity. The current YTM on the bonds is 4.6...
-
Dickson, Inc., has a debt-equity ratio of 2.3. The firms weighted average cost of capital is 9 percent and its pretax cost of debt is 5.3 percent. The tax rate is 24 percent. a. What is the companys...
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
A sample of ideal gas is in a sealed container. The temperature of the gas and the volume of the container are both increased. What other properties of the gas necessarily change? (More than one...

Study smarter with the SolutionInn App


