Here is the elemental composition (wt %) of a typical iron ore: Element Concentration7% Fe 59.2...
Fantastic news! We've Found the answer you've been seeking!
Question:
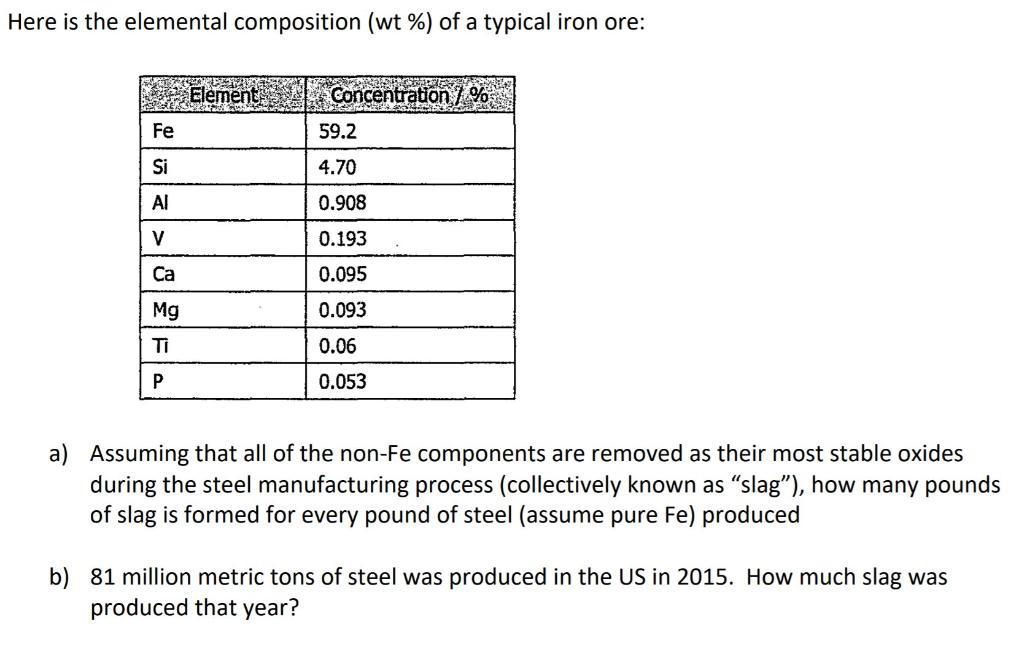
Transcribed Image Text:
Here is the elemental composition (wt %) of a typical iron ore: Element Concentration7% Fe 59.2 Si 4.70 Al 0.908 V 0.193 Ca 0.095 Mg 0.093 Ti 0.06 0.053 a) Assuming that all of the non-Fe components are removed as their most stable oxides during the steel manufacturing process (collectively known as "slag"), how many pounds of slag is formed for every pound of steel (assume pure Fe) produced b) 81 million metric tons of steel was produced in the US in 2015. How much slag was produced that year? Here is the elemental composition (wt %) of a typical iron ore: Element Concentration7% Fe 59.2 Si 4.70 Al 0.908 V 0.193 Ca 0.095 Mg 0.093 Ti 0.06 0.053 a) Assuming that all of the non-Fe components are removed as their most stable oxides during the steel manufacturing process (collectively known as "slag"), how many pounds of slag is formed for every pound of steel (assume pure Fe) produced b) 81 million metric tons of steel was produced in the US in 2015. How much slag was produced that year?
Expert Answer:
Answer rating: 100% (QA)
a Lets take the basis as 100 pounds of iron ore according to the data given Fe 592 ... View the full answer

Related Book For 

Mathematical Applications for the Management Life and Social Sciences
ISBN: 978-1305108042
11th edition
Authors: Ronald J. Harshbarger, James J. Reynolds
Posted Date:
Students also viewed these accounting questions
-
The three most stable oxides of carbon are carbon mono-oxide (CO), carbon dioxide (CO2), and carbon suboxide (C3O2). The molecules can be represented as Explain how these molecules illustrate the law...
-
The three most stable oxides of carbon are carbon monoxide (CO), carbon dioxide (CO2), and carbon suboxide (C3O2). The space- filling models for these three com-pounds are For each oxide, draw the...
-
The most important oxides of iron are magnetite,Fe3O4,and hematite,Fe 3O4 (a) What are the oxidation states of iron in these compounds? (b) One of these iron oxides is ferrimagnetic, and the other is...
-
Chris Co. purchased a piece of equipment and incurred the following costs. Which of these costs is properly capitalized and recorded as part of the cost of the equipment? I. Freight charges to...
-
These data track monthly performance of stock in Apple Computer since 1990. The data include 264 monthly returns on Apple Computer, as well as returns on the entire stock market, the S&P 500 index,...
-
Identify the potential barriers (or obstacles) that face companies considering or expanding international marketing operations. Which are the most important and which are less important? Explain.
-
Let \(f(\theta)\) be the \(2 \pi\)-periodic function determined by the formula \[f(\theta)=\theta^{2}, \quad \text { for }-\pi \leq \theta \leq \pi\] Find the Fourier series for \(f(\theta)\).
-
Compute the rate of return for the following cash flow to within 0.5%. Year Cash Flow 0..............................-$640 1.....................................0...
-
141 Camauts the activity rate for nach activity cast neal and Required information Machine setup Special processing General factory $ 1,000.00 per setup $ 10.00 per MH $ 63.00 per DLH a(2) Determine...
-
Three years ago. Karen Suez and her brother-in-law Reece Jones opened Glgasales Department Store. For the first 2 years, business was good, but the following condensed income statement results for...
-
Solve the inequality: 1-2126
-
Assuming general initial conditions, express the system model in a. Configuration form. b. Standard, second-order matrix form....
-
\(\frac{2 s+3}{\left(s^{2}+4 ight)(s+1)}\) Find the inverse Laplace transform by using the partialfraction expansion method.
-
Holyfield Corporation wishes to exchange a machine used in its operations. Holyfield has received the following offers from other companies in the industry. 1. Dorsett Company offered to exchange a...
-
The simplicity of the result is intuitive. Quite obviously, as one can see in the definition (7.3), the symmetry of the problem is much larger than the sole rotation in \(\mathcal{R}^{3}\). There is...
-
Table 18.3 gives some distances and commute times for a typical daily commute. Find the average speed (to the nearest mph) for the time intervals requested in Problems 1-4. Table 18. 3 \(6: 09\) to...
-
Based on the data provided below, calculate the charges for disposal of the waste with and without recycling at the transfer station, and comment on these costs Waste generated ...
-
Using the parallel-axis theorem, determine the product of inertia of the area shown with respect to the centroidal x and y axes. 6 in. 9 in. 9 in- 4.5 in. in. 4.5 in.
-
Solve each system of equations by using inverse matrices. 1. 2. 3. + 2z = 5 x + 4y 23D 2 + = -3 + + 22 - -x+ 4y + 2z = -10 2 + 5y + 3z -6
-
A bag contains 6 red balls and 4 black balls. If we draw 5 balls, with each one replaced before the next is drawn, what is the probability that at least 2 balls drawn will be red?
-
Use the definition of derivative to find f(x) for f (x) = 3x2 + 2x - 1.
-
What is the change in velocity of \((a)\) cart 1 (b) cart 2 in Figure 4.6? (c) What do you notice about your two answers? Figure 4.6 Velocity-versus-time graph for two identical carts before and...
-
(a) Are the accelerations of the motions shown in Figure 4.1 constant? (b) For which surface is the acceleration largest in magnitude? Figure 4.1 Velocity-versus-time graph for a wooden block sliding...
-
The \(x\) component of the final velocity of the standard cart in Figure 4.8 is positive. Can you make it negative by adjusting this cart's initial speed while still keeping the half cart initially...

Study smarter with the SolutionInn App


