How many atoms are present in each of the following samples? a) 0.386 mol of HO...
Fantastic news! We've Found the answer you've been seeking!
Question:

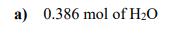
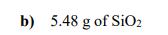
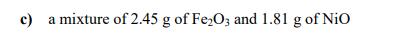
Transcribed Image Text:
How many atoms are present in each of the following samples? a) 0.386 mol of H₂O b) 5.48 g of SiO₂2 c) a mixture of 2.45 g of Fe₂O3 and 1.81 g of NiO How many atoms are present in each of the following samples? a) 0.386 mol of H₂O b) 5.48 g of SiO₂2 c) a mixture of 2.45 g of Fe₂O3 and 1.81 g of NiO
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many rings are present in each of the following compounds? Each consumes 2 moles of hydrogen on catalytic hydrogenation. (a) C8H8 (b) C8H8CI2 (c) C8H8O (d) C8H10O2 (e) C8H9ClO
-
How many asymmetric carbon atoms are present in each of the following compounds? (a) (b) (c) H-C-C-C-CI H CI H OH CH CH2OH OH H H OH
-
How many unpaired electrons are present in each of the following in the ground state: O, O+, O-, Os, Zr, S, F, Ar?
-
Write each number in scientific notation. 0.875
-
Three years ago, you founded your own company. You invested $100,000 of your own money and received 5 million shares of Series A preferred stock. Your company has since been through three additional...
-
The inductors in Fig. 6.87 are initially charged and are connected to the black box at t = 0. If i1(0) = 4 A, i2(0) = -2 A, and v(t) = 50e-200t mV, t ¥ 0$, find: (a) The energy initially stored...
-
Use the quotient rule to simplify \(\frac{b^{6}}{b^{4}}\).
-
Chapman Department Store is located in midtown Metropolis. During the past several years, net income has been declining because suburban shopping centers have been attracting business away from city...
-
The following inventory balances have beenprovided for the mostrecent year:Raw materials, Beginning-Raw materials, Ending Work in process,beginning Work in process, Ending-Finished goods,Beginning 2...
-
You work in the human resources department of your company helping new employees fill out the necessary paperwork to get their first paycheck. There are a number of decisions that employees must make...
-
A conpany wants to grow within its existing market. What is this strategy called and what are 3 suitable modes of growth for them to consider: what are pros and cons of each
-
Starting on her 40 th birthday, Lynette plans to start saving for her retirement.She will contribute $8,000 to a brokerage account each year on her birthday, starting today. Her 25 th and final...
-
Research and graph bar charts comparing the following aspects about The New Deal and the Stimulus Package: Effect on unemployment rate Estimated number of jobs created as a percentage of the...
-
An arrow fired horizontally at 35m/s travels 27mhorizontally, before landing on the ground. a) From what height above the ground was it fired? Express your answer to two significant figures and...
-
Thank you for applying for Brand Associate position at Gap Factory Highstreet. My name is Avis, I am the General Manager. I would love to connect with you about this position. This email is the first...
-
ABN Ltd shares have a closing price of $10.85 on 8 November 2005. On the next day they will begin trading on an ex-dividend basis. The dividend is 40 cents per share fully franked at the company tax...
-
help to research two journal articles from the APUS online library on the same reverse logistics topic and writing a summary of each article comparing both in terms of(1)howeach definedtheir specific...
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
Sodium hydroxide is hygroscopic-that is, it absorbs moisture when exposed to the atmosphere. A student placed a pellet of NaOH on a watch glass. A few days later, she noticed that the pellet was...
-
A phase diagram of water is shown at the end of this problem. Label the regions. Predict what would happen as a result of the following changes: (a) Starting at A, we raise the temperature at...
-
Assuming that air contains 78 percent N2, 21 percent O2, and 1 percent Ar, all by volume, how many molecules of each type of gas are present in 1.0 L of air at STP?
-
Paymore Shoes acquired 80 percent of the voting stock of Spire Footwear on February 1, 2014, for \($21\) million. The fair value of the noncontrolling interest at the acquisition date was \($3\)...
-
Pacific Athletic Corporation owns all of the voting stock of Solovair Apparel. Acquisition cost was \($10\) million in excess of Solovairs book value of \($2\) million, and the excess was attributed...
-
Peninsula Industries and Seaport Company, a 90 percent owned subsidiary, engage in extensive intercompany transactions involving raw materials, component parts, and completed products. Peninsula...

Study smarter with the SolutionInn App



