How many grams of sucrose (C12H22O11) are in 1.55 L of 0.758 M sucrose solution? Express...
Fantastic news! We've Found the answer you've been seeking!
Question:
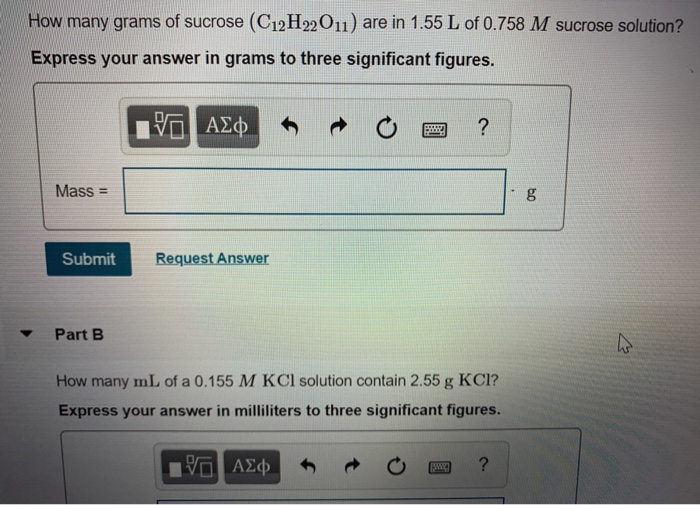
Transcribed Image Text:
How many grams of sucrose (C12H22O11) are in 1.55 L of 0.758 M sucrose solution? Express your answer in grams to three significant figures. 17 ΑΣΦ Mass - Submit Request Answer Part B ? How many mL of a 0.155 M KCl solution contain 2.55 g KCI? Express your answer in milliliters to three significant figures. — ΑΣΦ ? to g How many grams of sucrose (C12H22O11) are in 1.55 L of 0.758 M sucrose solution? Express your answer in grams to three significant figures. 17 ΑΣΦ Mass - Submit Request Answer Part B ? How many mL of a 0.155 M KCl solution contain 2.55 g KCI? Express your answer in milliliters to three significant figures. — ΑΣΦ ? to g
Expert Answer:
Answer rating: 100% (QA)
Answer to part A To answer this question we first need to find two things that has not been given in ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many grams of sucrose (C12H22O11, molar mass: 342.29 g/mol) are needed to lower the freezing point of 100 g of water by 25.00 C? (Kf for water is 1.86 C/m) a) 2.912 g b) 955 g c) 10.5 g d) 459.7...
-
How many grams of matter would have to totally destroy to run a 100-W lightbulb for 1 year?
-
How many grams of N2 will react if 100.0 kJ of energy are supplied? N2(g) + O2(g) ( 2NO(g)H = 180.6 kJ
-
Describe, in human terms, why delay and jitter are bad in real time (interactive) voice and video communications. Would these same problems apply to recorded voice and video stored and played back at...
-
What is securitization? What forces underlie it, and how has it affected the financing policies of multinational corporations?
-
In its annual report, BALLI Supply includes the following five-year financial summary: Requirement 1. Analyze the companys financial summary for the fiscal years 20082012 to decide whether to invest...
-
1. Identify an entrepreneur in your area you would like to interview. 2. Contact the person you have selected and make an appointment. Be sure to explain why you want the appointment and to give a...
-
CVP analysis, shoe stores. The WalkRite Shoe Company operates a chain of shoe stores that sell 10 different styles of inexpensive men's shoes with identical unit costs and selling prices. A unit is...
-
A tiling company has been tasked to tile the floor of a new shopping mall using the following materials and creating the design which follows: . Square marble slabs . Heavy duty rods on the outer...
-
CableTech Bell Corporation (CTB) operates in the telecommunications industry. CTB has two divisions: the Phone Division and the Cable Service Division. The Phone Division manufactures telephones in...
-
Can not figure out what is wrong with my Java code private char suit; private int value; //+PlayingCard (s;char, v;int) public playingCard(char s, int v){ suit = s; value = v; } //get suit public...
-
What best practices could be instituted to manage HIPAA compliance while aligning with the organization's mission and ethical standards?
-
think about an organizational improvement initiative that you would like to make in an organization of your choice. This initiative might focus on improved profitability, better customer service, new...
-
in Pavan Sukhdev's video "Put Value on Nature", he calls for change in our accounting systems to truly account for the costs of production. Do you think this is realistic? What value would obtain...
-
An employee is given a $10,000 interest free loan from an employer on January 1 to purchase an automobile to be used for employment purposes. Due to a serious illness, the automobile is only used for...
-
Ally, the manager of a disability services agency in regional Queensland, is a qualified social worker with 10 years of managerial experience. The agency she manages provides accommodation and...
-
Given the following factored polynomial, p(x)=x(2-x)(x+2)5 Perform the Multiplicity Test by answering the following: The zero x= -2 has multiplicity A/ The zero x= 0 has multiplicity The zero x= 2...
-
Use the T account for Cash below to record the portion of each of the following transactions, if any that affect cash. How do these transactions affect the companys liquidity? Jan. 2 Provided...
-
The first step in the industrial recovery of zinc from the zinc sulfide ore is roasting, that is, the conversion of ZnS to ZnO by heating: 2ZnS(s) + 3O2(g) 2ZnO(s) 2SO2(g) (H = -879 kJ/mol Calculate...
-
Hydrogen halides (HF, HCl, HBr, HI) are highly reactive compounds that have many industrial and laboratory uses. (a) In the laboratory, HF and HCl can be generated by reacting CaF2 and NaCl with...
-
Consider the following two reactions: Determine the enthalpy change for the process 2B - C A 2B
-
Because of the negative incentive effect that taxes have on goods with elastic supply, in the late 1980s Margaret Thatcher (then prime minister of Great Britain) changed the property tax to a poll...
-
Many of the buildings in Paris have Mansard roofs, such as those shown in the photograph on page 147. a. What property tax structure would bring this about? b. Could you imagine a change in the...
-
On September 29, 2008, the stock market fell almost 7%. How can we reconcile these kinds of huge losses in the stock market with the efficient market hypothesis?

Study smarter with the SolutionInn App


