How many moles of K are there in a sample of K that contains 1.14x1024 atoms?...
Fantastic news! We've Found the answer you've been seeking!
Question:
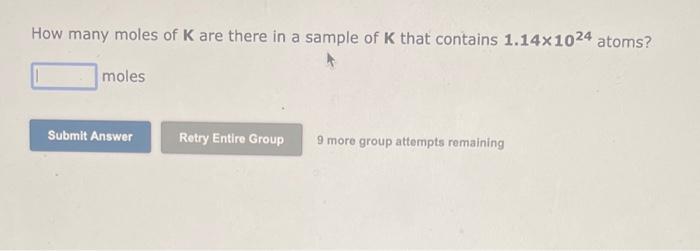
Transcribed Image Text:
How many moles of K are there in a sample of K that contains 1.14x1024 atoms? moles Submit Answer Retry Entire Group 9 more group attempts remaining How many moles of K are there in a sample of K that contains 1.14x1024 atoms? moles Submit Answer Retry Entire Group 9 more group attempts remaining
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many moles and molecules of K are there in a 27 g sample of KCIO? Knowing that it contains 76 g of the compound, determine the percentage of K in the sample.
-
How many moles of BH3 are needed to react with 2 moles of 1-pentene?
-
How many moles of O2 are needed to prepare 1.00 g of Ca(NO3)2? Ca(s) + N2(g) + 3O2(g) ( Ca(NO3) 2(s)
-
CMOS Chips is hedging a 20-year, $21 million, 8% bond payable with a 20-year interest rate swap and has designated the swap as a fair value hedge. The agreement called for CMOS to receive payment...
-
Assume that on January 1, 2014, an Early Risers Caf purchased a building, paying $54,000 cash and signing a $104,000 note payable. The restaurant paid another $65,000 to remodel the building....
-
Medway Printers (MP) manufactures printers. Assume that MP recently paid $900,000 for a patent on a new laser printer. Although it gives legal protection for 20 years, the patent is expected to...
-
The Thermo-Bond Manufacturing Company maintains its fixed-asset records on its computer. The fixed-asset master file includes the following data items: Required Refer to Table 9-7, which describes...
-
U.S. Multinational Corporations subsidiary in Bangkok has on its books fixed assets valued at 7,500,000 baht. One-third of the assets were acquired two years ago when the exchange rate was THB40 =...
-
31 December 2022 - calculate the fuel tax credits to be claimed Chart of accounts is below to know what accounts to debit and credit for question 2. Please include narration in manual journal. 1....
-
Two power plants are currently emitting 8,000 tons of pollution each (for a total of 16,000 tons) in City A. Marginal pollution reduction costs for Plant 1 are given by MCR 1 = 0.02 Q 1 and for Plant...
-
A distributor sells clothing for retailers and agrees to take back the units not sold without any discount, an incentive for the retailers to order more. The market has five geographical areas, each...
-
Which of the following is a signal that a company may be using SPEs to overstate its earnings or understate its debt? (a) When references in the notes to the financial statements regarding...
-
On your own or with a classmate, choose one of the following products and create an advertisement that illustrates how your company creates time, place, and form utility in its delivery of the...
-
The Secondary Mortgage Market Enhancement Act of 1984: (a) Allowed the GSEs (Fannie Mae and Freddie Mac) to sell MBSs (mortgagebacked securities) backed by FHA and VA loans. (b) Allowed institutional...
-
Signal #1 for the scheme of improper use of acquisition or merger reserves is a parent company allocating large amounts to goodwill when acquiring other companies. Explain why this could be a signal...
-
How would you decide whether to use an investment advisor or stockbroker at a full-service brokerage firm such as Charles Schwab or to open a brokerage account and try your hand at do-it-yourself...
-
Planning Project Office on the web Frame Requirement Analysis Definition of Technologies and Methods Conceptualization Project Design * Software Design Status Report Implementation Integration User...
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
Describe how you would prepare astatine-211, starting with bismuth-209.
-
Acetic acid (CH 3 COOH) is an important ingredient of vinegar. A sample of 50.0 mL of commercial vinegar is titrated against a 1.00 M NaOH solution. What is the concentration (in M ) of acetic acid...
-
Calculate the concentration of OH- ions in a 1.4 3 1023 M HCl solution.
-
The Counter Company just completed the year-end physical count of its inventory. The total value of inventory was determined to be \($300,000\). The following additional information came to light...
-
Williams Distributing Company is a merchandising company. Williams uses the perpetual inventory system. eames, Record each of the following transactions related to the companys purchasing and selling...
-
On June 15, a customer buys 75 CDs on account from Musicland for \($10\) each, for a total of \($750\). The list price of the CDs is \($12\) each, but Musicland gave the customer a \($2\) per CD...

Study smarter with the SolutionInn App



