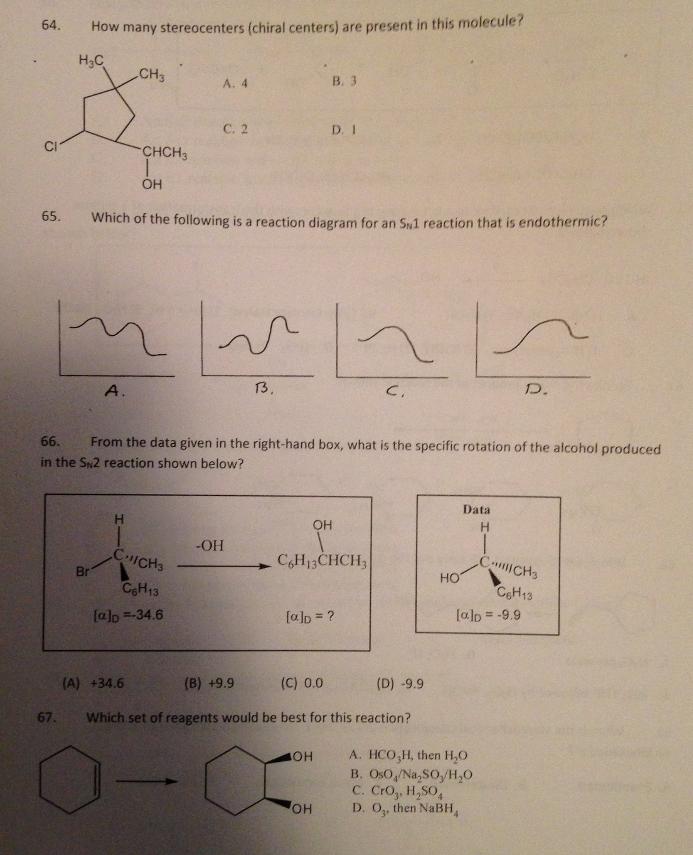
64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO,
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Which of the following is equivalent to u=-Cost+V3 sint ? a) u = 2 cos t + 3 1 b) u = - cost+ 3 c) u = 2 cos t 3 d) u = : 1 os t- 3 tho
-
Which of the following is not related to ethical requirements of auditors? a. Due care. b. Independence in appearance. c. Independence in fact. d. Professional judgment.
-
Which of the following is not required by generally accepted auditing standards? a. Written representations. b. Attorney letter. c. Management letter. d. Engagement letter.
-
Which of the following pairs of steps in the programming process is in the correct order? a. Code the program, plan the logic b. Test the program, translate it into machine language c. Put the...
-
Refer to the transactions described in P24A. Keep in mind that Jake may not need to record all transactions. Required: 1. Record each transaction for Lukes Repair Shop. Keep in mind that Luke may not...
-
Solkis Repair Shop was started on May 1 by Solki Lee. A summary of May transactions is presented below. 1. Invested $10,000 cash to start the repair shop. 2. Purchased equipment for $5,000 cash. 3....
-
Suppose you're evaluating a stock, and you expect the next dividend to be $0.57, you expect future dividends on this stock to increase at a rate of 2 percent per quarter, and you think the...
-
In the early 1970s a widely publicized list of the "Nifty Fifty" stocks was drawn up. This list, which included Avon Products, Polaroid, Coca-Cola, McDonald's, Walt Disney, American Express, and...
-
ct one: a. m2BAC = 90 b. m BAC 30 c. m2BAC = 45 d. m2BAC = 60 The vertices of a triangle are as follows: A = (3,-2), B = (7,-2), and C = (7, -6). What is the m2BAC = ? Here is the link to Geogebra.
-
Use the data in MURDER.RAW for this exercise. The variable mrdrte is the murder rate, that is, the number of murders per 100,000 people. The variable exec is the total number of prisoners executed...
-
Question 3 104 (5 marks) Complete the chart below by placing an X in the approp Type o Sole Characteristic Proprietorship Limited liability Unlimited liability X Owners are shareholders Owners are...
-
Simplify: (3xy) (4x)
-
Discuss aspects of number theory related to prime numbers and discrete logarithms ?
-
3. A solid uniform semi-circle with radius R is placed along the x axis, with its axis of symmetry along y axis. (a) Where is the center of mass of the semi-circle, in coordinate form? (b) If the...
-
Ivana invited Yola to her house on Saturday at four o'clock pm for a catfish and hush puppy dinner, and Yola agreed to come. Yola arrives on Saturday on time, only to find that Ivana left town and...
-
What is financial leverage and how can it impact a business?
-
The following table contains information about four potential investment projects that Castle Corporation is considering Project Required Investment Project Life Payback Acctg ROR Period NPV...
-
Burberrys competitive advantage is through its differentiation strategy. What risk should Burberry remain aware of?
-
Farha Co. purchases a machine for $11,500, terms 2/10, n/60, FOB shipping point. The seller pre paid the $260 freight charges, adding the amount to the invoice and bringing its total to $11,760. The...
-
On April 1, 2004, Stones Backfioe Co. purchases a trencher for $250,000. The machine is expected to last five years and have a salvage value of $25,000. Compute depreciation expense for year 2005...
-
On January 1, KB Store had $450,000 of inventory at cost. In the first quarter of the year, it purchased $1,590,000 of merchandise, returned $23,100, and paid freight charges of $37,600 on purchased...

Study smarter with the SolutionInn App


