How many units did the client produce last year? Linda is studying the working papers from her
Fantastic news! We've Found the answer you've been seeking!
Question:
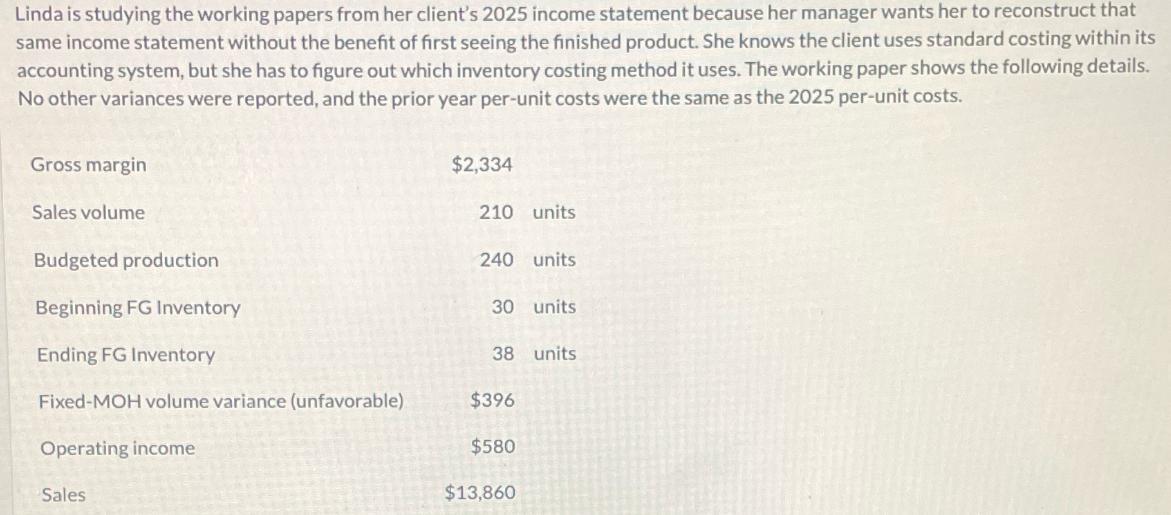
Transcribed Image Text:
Linda is studying the working papers from her client's 2025 income statement because her manager wants her to reconstruct that same income statement without the benefit of first seeing the finished product. She knows the client uses standard costing within its accounting system, but she has to figure out which inventory costing method it uses. The working paper shows the following details. No other variances were reported, and the prior year per-unit costs were the same as the 2025 per-unit costs. Gross margin Sales volume Budgeted production Beginning FG Inventory Ending FG Inventory Fixed-MOH volume variance (unfavorable) Operating income Sales $2,334 210 units 240 units 30 units 38 units $396 $580 $13,860 Linda is studying the working papers from her client's 2025 income statement because her manager wants her to reconstruct that same income statement without the benefit of first seeing the finished product. She knows the client uses standard costing within its accounting system, but she has to figure out which inventory costing method it uses. The working paper shows the following details. No other variances were reported, and the prior year per-unit costs were the same as the 2025 per-unit costs. Gross margin Sales volume Budgeted production Beginning FG Inventory Ending FG Inventory Fixed-MOH volume variance (unfavorable) Operating income Sales $2,334 210 units 240 units 30 units 38 units $396 $580 $13,860
Expert Answer:
Answer rating: 100% (QA)
To determine the number of units produced last year we need to calculat... View the full answer

Related Book For 

Auditing An International Approach
ISBN: 978-1259087462
7th edition
Authors: Wally J. Smieliauskas, Kathryn Bewley
Posted Date:
Students also viewed these accounting questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Suppose a state was trying to decide whether to fund primary and secondary education with a property tax or an income tax. What implications might this choice have for land use in the state?
-
Two horses pull horizontally on ropes attached to a stump. The two forces F1, and F2 that they apply to the stump are such that the net (resultant) force R has a magnitude equal to that of F, and...
-
What is apparent from a horizontal presentation of financial statement information? A vertical presentation?
-
What are the features of the US Toxic Release Inventory that other ecological reporting systems should consider adopting? Are there any problems with the TRI reporting system? If so, how can these be...
-
A corporation issues $18,000,000 of 10% bonds to yield interest at the rate of 8%. (a) Was the amount of cash received from the sale of the bonds greater or less than $18,000,000? (b) Identify the...
-
Hero X DZL Homepage - Central State Univer x + content/23319/viewContent/1056574/View Time Taken:0:00:54 Austin Green: Attempt 1 Question 3 (1 point). Listen Provide an appropriate response. Find the...
-
Regulators, in protecting the public interest, are examining Wincox Insurance Company's financial statements. These regulators impose requirements on Wincox's financial statements because of which...
-
A solution consists of 54.2 g of silver nitrate, AgNO3, and 83.8g water and has a density of 1.46 g/mL. Calculate the weightpercent of AgNO3, the molality of AgNO3, the mole fraction ofAgNO3, and the...
-
List some of the testing patterns that can be applied for testing the corruption analysis pattern.
-
T/F: developing conceptual models requires both domain knowledge and modeling skills.
-
What do you think are the implementation issues for the AnyReason BO, when used with the corruption pattern?
-
Are certain modifications required in our stability model so that even the anomalies can fit in?
-
What do you think are the implementation issues for the AnyEvidence BO, when used in the corruption analysis pattern?
-
(i) Derive IS curve and draw it (12 points) (ii) What is the change in net export after an increase in goverment expen- diture ? (12 points)
-
A sample statistic will not change from sample to sample. Determine whether the statement is true or false. If it is false, rewrite it as a true statement.
-
What is the audit trail? What is its use in the audit?
-
Two aspects that affect the form of opinion in an auditors report are a. The need to issue a modified report or an adverse report. b. The scope of the auditors work and the fairness of presentation...
-
What are some common substantive audit procedures for revenues, cash, and accounts receivable?
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
A system is said to be in thermodynamic equilibrium if its (a) Temperature remains unchanged (b) Pressure remains unchanged (c) Chemical potential remains unchanged (d) Temperature, pressure and...
-
A system initially containing \(3 \mathrm{~mol} \mathrm{CO}_{2}, 5 \mathrm{~mol} \mathrm{H}_{2}\) and \(1 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\) is undergoing the following reactions: \[...

Study smarter with the SolutionInn App


