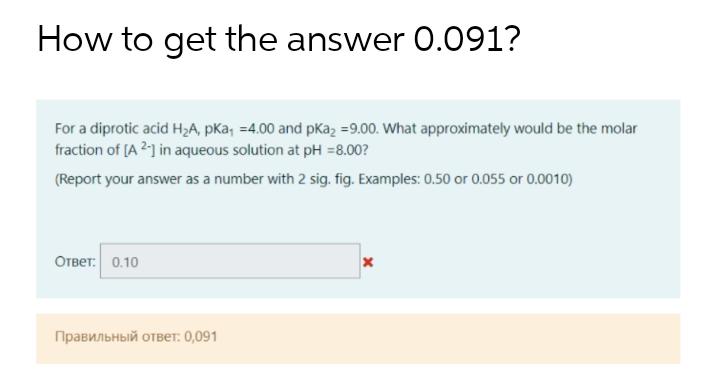
How to get the answer 0.091? For a diprotic acid HA, pKa, -4.00 and pka =9.00....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
How to get the answer 0.091? For a diprotic acid H₂A, pKa, -4.00 and pka₂ =9.00. What approximately would be the molar fraction of [A2-] in aqueous solution at pH = 8.00? (Report your answer as a number with 2 sig. fig. Examples: 0.50 or 0.055 or 0.0010) Ответ:0.10 Правильный ответ: 0,091 x How to get the answer 0.091? For a diprotic acid H₂A, pKa, -4.00 and pka₂ =9.00. What approximately would be the molar fraction of [A2-] in aqueous solution at pH = 8.00? (Report your answer as a number with 2 sig. fig. Examples: 0.50 or 0.055 or 0.0010) Ответ:0.10 Правильный ответ: 0,091 x
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
The mobility of a CH1COi ion in aqueous solution at 25C is 4.24 x 10-8 m2 S-1 V-1 Calculate its diffusion coefficient in water at 25C.
-
A typical diffusion coefficient for small molecules in aqueous solution at 25C is 5 10 9 m 2 s 1 . If the critical reaction distance is 0.4 nm, what value is expected for the second-order rate...
-
The mobility of an acetate ion in aqueous solution at 25C is 4.24 x 10-8 m3 S-1 V-1. Calculate the molar ionic conductivity.
-
A person is pulling on a rope attached to a locomotive of mass m = 100 tons (Fig. 1, right). The person is pulling with a constant force F that is 2 times greater than their body weight, at an upward...
-
Stone Sour Corp. issued 20-year bonds two years ago at a coupon rate of 7.1 percent. The bonds make semiannual payments. If these bonds currently sell for 105 percent of par value, what is the YTM?
-
A firms bonds have a maturity of 8 years with a $1,000 face value, have an 11% semiannual coupon, are callable in 4 years at $1,154, and currently sell at a price of $1,283.09. What are their nominal...
-
The angular momentum of a hydrogen atom is \(4.70 \times 10^{-34} \mathrm{~J} \cdot \mathrm{s}\). What is the minimum energy, in \(\mathrm{eV}\), that this atom could have?
-
Hills operations manager (see Problems 13.3 through 13.5) is also considering two mixed strategies for January-August: (a) Plan D: Keep the current workforce stable at producing 1,600 units per...
-
Design test cases to achieve loop coverage on the function "computeTax" (list the loops covered by each test case). If it is not feasible, explain the reason. Try your best to use the minimum number...
-
As loan analyst for Murray Bank, you have been given the following information. Each of these companies has requested a loan of 50,000 for 6 months with no collateral offered. Since your bank has...
-
The following information relates to a joint production process for three products, with a total joint production cost of $135,000. There are no separable processing costs for any of the three...
-
n what ways do globalization and technological advancements impact social identities and cultural practices, and how do individuals and communities negotiate their sense of belonging and cultural...
-
ACC 111 Project Information - Project 1 - Why Accounting/Business/Human Resources, etc. This Project will consist of a discussion of why you chose your current major. In your conclusion, state the...
-
2.4. What is the intensity of radiation emitted by a hot desert (330 K) relative to that emitted by the strato- sphere over the South Pole during July (190 K)?
-
a) b) Ic) d) Gilbert holds the DCO Canadian Growth Segregated Fund. After flat fund performance in the first 3 months of the year, he decides to switch to the DCO U.S. Growth Segregated Fund. Which...
-
Firm F needs a new truck. It has two opportunities: either lease or buy. The lease contract is 4 years. The before-tax lease payment is 15,000 per year (claimed at the beginning of the year). If firm...
-
Selma and Troy are planning to take a second honeymoon in 2 years. They estimate that the cost will be roughly $7,985.74. They plan on making monthly payments of $275 and expect to receive 10% annual...
-
What services are provided by the provincial and territorial governments?
-
What are the principal advantages and the principal limitations of each of the detectors listed in Problem 32-10? In Problem 32-10 Describe the principle on which each of the following GC detectors...
-
What minimum requirement is needed to obtain reproducible results with a single-beam spectrophotometer?
-
A volumetric calcium analysis on triplicate samples of the blood serum of a patient believed to be suffering from a hyperparathyroid condition produced the following data: mmol Ca/L = 3.15, 3.25,...
-
The descending order in which current assets should be shown in the statement of financial position is (A) Inventory, Accounts receivable. Bank, Cash (B) Cash, Bank, Accounts receivable, Inventory...
-
Which of these best describes non-current assets? (A) Items bought to be used in the business (B) Items which will not wear out quickly (C) Expensive items bought for the business (D) Items having a...
-
The costs of putting goods into a saleable condition should be charged to (A) The trading account (B) The profit and loss account (C) The statement of financial position (D) None of these

Study smarter with the SolutionInn App



