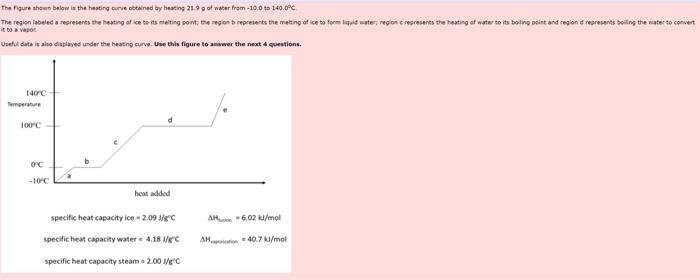
The figure shown below is the heating curve obtained by heating 21.9 g of water from-10.0...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
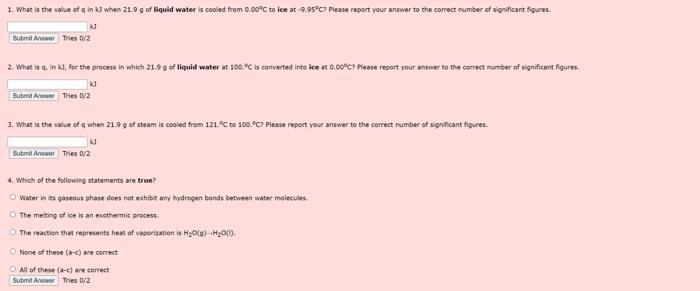
The figure shown below is the heating curve obtained by heating 21.9 g of water from-10.0 to 140.0°C. The region labeled a represents the heating of ice to its melting point; the region b represents the metting of ice to form liquid water; region c represents the heating of water to its boiling point and region d represents boiling the water to convert it to a vapor Useful data is also displayed under the heating curve. Use this figure to answer the next 4 questions. 140°C Temperature 100°C- OFC -10°C heat added specific heat capacity ice-2.09 1/g°C specific heat capacity water 4.18 1/gC specific heat capacity steam = 2.00 1/g"C AH-6.02 kJ/mol AH-40.7 kJ/mol 1. What is the value of q ink when 21.9 g of liquid water is cooled from 0.00°C to ice at -9.95°C? Please report your answer to the correct number of significant figures. kd Submit Answer Tries 0/2 2. What is g. in k3, for the process in which 21.9 g of liquid water at 100 °C is converted into ice at 0.00°C? Please report your answer to the correct number of significant figures. kd Submit Answer Tries 0/2 3. What is the value of q when 21.9 g of steam is cooled from 121.°C to 100.9C7 Please report your answer to the correct number of significant figures. kd Submit Anwer Tries 0/2 4. Which of the following statements are true? Water in its gaseous phase does not exhibit any hydrogen bonds between water molecules The melting of ice is an exothermic process. The reaction that represents heat of vaporization is H₂O(g) +H₂O(1) None of these (a-c) are correct All of these (a-c) are correct Submit Answer Tries 0/2 The figure shown below is the heating curve obtained by heating 21.9 g of water from-10.0 to 140.0°C. The region labeled a represents the heating of ice to its melting point; the region b represents the metting of ice to form liquid water; region c represents the heating of water to its boiling point and region d represents boiling the water to convert it to a vapor Useful data is also displayed under the heating curve. Use this figure to answer the next 4 questions. 140°C Temperature 100°C- OFC -10°C heat added specific heat capacity ice-2.09 1/g°C specific heat capacity water 4.18 1/gC specific heat capacity steam = 2.00 1/g"C AH-6.02 kJ/mol AH-40.7 kJ/mol 1. What is the value of q ink when 21.9 g of liquid water is cooled from 0.00°C to ice at -9.95°C? Please report your answer to the correct number of significant figures. kd Submit Answer Tries 0/2 2. What is g. in k3, for the process in which 21.9 g of liquid water at 100 °C is converted into ice at 0.00°C? Please report your answer to the correct number of significant figures. kd Submit Answer Tries 0/2 3. What is the value of q when 21.9 g of steam is cooled from 121.°C to 100.9C7 Please report your answer to the correct number of significant figures. kd Submit Anwer Tries 0/2 4. Which of the following statements are true? Water in its gaseous phase does not exhibit any hydrogen bonds between water molecules The melting of ice is an exothermic process. The reaction that represents heat of vaporization is H₂O(g) +H₂O(1) None of these (a-c) are correct All of these (a-c) are correct Submit Answer Tries 0/2
Expert Answer:
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemical engineering questions
-
A hygrometer is used to measure the moisture content of humid air. Calibration of the instrument leads to a straight line on a semi log plot of y, the mole fraction of water in air (logarithmic...
-
The moisture content of a soil sample is 17% and the dry unit weight is 16.51 kN/m3. If Gs = 2.69, what is the degree of saturation?
-
A Venturi meter can be used to measure the flow speed of a liquid. A simple such device is shown in Fig. 9.44. Show that the flow speed of an ideal fluid is given by 2gAh 1- (AA) - 1 A1 A, Az
-
Discuss what you see as the role of ethics as it pertains to management and managers. Does management, in your view, help shape the values and ethics of an organization? What about an organization's...
-
Many people believe that students gain weight as freshmen. Suppose we plan to conduct a study to see if this is true. a) Describe a study design that would require a matched-pairs t procedure to...
-
A Ping-Pong ball weighs 2.6 g and has a diameter of 3.8 cm. It can be supported by an air jet from a vacuum cleaner outlet, as in Fig. P7.84. For sea level standard air, what jet velocity is required
-
The efficiency of a self-locking machine is _____ 50 percent.
-
Define the phrase earnings management. Under what conditions, if any, is earnings management acceptable? Do auditors responsibilities include actively searching for instances of earnings management...
-
Identify and describe a specific situation you have faced in the past, that involves a conflict, and difficult conversation. Discuss strategies you could employ in this situation that would enhance...
-
Ben Swanson, owner and manager of Swansons Department Store, has decided to use statistical forecasting to get a better handle on the demand for his major products. However, Ben now needs to decide...
-
You have your RHIT degree and have been working as a coder for 3 years. It is time for you to pursue a job advancement with the potential for a wage increase. You have the experience but realize that...
-
Jessica paid estimate tax payments for 2019 as follows: Date of Payment 4/15/2019 6/15/2019 9/15/2019 1/15/2020 Federal $2,000 $2,000 $2,000 $2,000 State $ 300 $ 400 $0 $ 300 Prepare Jessica's 2019...
-
Find the slope-intercept form of the line with the given slope and y-intercept. 9 3 Slope = 2 y-intercept = 5 What is the equation of the line? (Simplify your answer. Type your answer in...
-
Interest income on loans and securities$12,000 Interest paid on deposits10,000 Non-interest income2,500 Total non-interest expense3,500 Provision for loan loss500 Security gains 250 Income taxes210...
-
The following C program is to be compiled by a compiler targeting the ARMv8-A architecture: long long int a = 10; long long int b = 29; long long int c =30; long long int d =0; void main() { d =...
-
Herman and Sons' Law Offices opened on January 1, 2022. Herman's adjusted trial balance at December 31, 2022 is as follows: (Click the icon to view the adjusted trial balance.) Requirements Perform...
-
Identify three to five factors (with cost categories) that contribute to the cost of quality. Explain how these factors are in place in a military organization? If unable to explain, how would you...
-
San Carlos Bank and Trust Company uses a credit-scoring system to evaluate most consumer loans that amount to more than $2,500. The key factors used in its scoring system are found at the conclusion...
-
Convert the temperatures in parts (a) and (b) and temperature intervals in parts (c) and (d): (a) T = 85F to R, C, K (b) T = 10C to K, F, R (c) T = 85C to K, F, R (d) T = 150R to F, C, K
-
A gas cylinder filled with nitrogen at standard temperature and pressure has a mass of 37.289 g. The same container filled with carbon dioxide at STP has a mass of 37.440 g. When filled with an...
-
Certain solid substances, known as hydrated compounds, have well-defined molecular ratios of water to some other species, which often is a slat. For example, calcium sulfate dehydrate (commonly known...
-
An analysis by Professor Peter M Rothwell and his colleagues (Nuffield Department of Clinical Neuroscience, University of Oxford, UK) published in 2012 in the medical journal The Lancet...
-
The following table shows the result of the 2018 General Elections in Pakistan, along with the vote share predicted by several organizations in the days before the elections. The sample sizes were...
-
The Chinese Academy of Sciences and the Chinese Academy of Environmental Planning conducted a survey of about 563 Chinese, with a tertiary education, about their interest in reducing global warming....

Study smarter with the SolutionInn App



