Hydroboration-oxidation of (E)-2-(p-anisyl)-2-butene yielded an alcohol A, mp 60 C, in 72% yield. When the same...
Fantastic news! We've Found the answer you've been seeking!
Question:
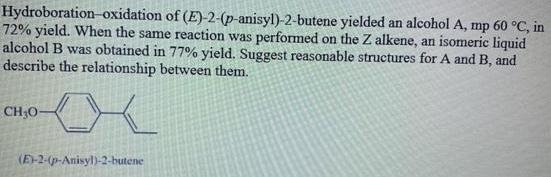
Transcribed Image Text:
Hydroboration-oxidation of (E)-2-(p-anisyl)-2-butene yielded an alcohol A, mp 60 °C, in 72% yield. When the same reaction was performed on the Z alkene, an isomeric liquid alcohol B was obtained in 77% yield. Suggest reasonable structures for A and B, and describe the relationship between them. CH;O- (E-2-(p-Anisyl)-2-butene Hydroboration-oxidation of (E)-2-(p-anisyl)-2-butene yielded an alcohol A, mp 60 °C, in 72% yield. When the same reaction was performed on the Z alkene, an isomeric liquid alcohol B was obtained in 77% yield. Suggest reasonable structures for A and B, and describe the relationship between them. CH;O- (E-2-(p-Anisyl)-2-butene
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A Chinese automobile company Geely Auto aspires to enter the US market by setting up their manufacturing unit there and producing cars locally to give competition to their US and European...
-
Suggest reasonable structures for compounds A, B, and C in the following reactions:
-
Suggest reasonable structures for compounds A, B, and C in the following reactions: Compound C is more stable than compound A. OTs stands for toluenesulfonate. CHIA-T-ms won.com posad A+ compound...
-
Violet Flowers expects to sell 3,000 plants a month. She estimated the following monthly costs: Variable Costs $9,000 Fixed Costs $15,000 During her second month of operation, Violet would like to...
-
Send an e-mail message to your instructor describing an ethical or legal communication that you had to handle at your job or in the classroom. Apply the ethical principles described in this chapter...
-
On March 1, 2017, Company "Z" had 1800 merchandise inventories at a cost of $59.90 each. The company uses periodic inventory system and has completed the following merchandising transactions during...
-
Patty Hayes owned four Personal Seat Licenses (PSLs) at the Cleveland Browns Stadium. Hayess PSLs reserved four seats on the 50-yard line, at the railing, on the north side of the stadium. As the...
-
In eight years, Kent Duncan will retire. He is exploring the possibility of opening a self-service car wash. The car wash could be managed in the free time he has available from his regular...
-
(a) Run linear regression on the closing price data for BTC in the range of 12/15/2020 to 12/28/2020. Report the r-squared value on your training data. Use this to predict BTC closing prices on...
-
The assets, labilities, and equities of Dundee Design Studio have the following balances at December 31, 2024. The Retained Earnings was $31,000 at the beginning of the year. At year end, Common...
-
The gas mileage of a subcompact car is given by m = -0.04x 2 + 3.6x - 49, where x = the speed in miles per hour and m is given in miles per gallon. a) What is the gas mileage for a speed of 25 miles...
-
Which steps normally establish an implied contract?
-
Ordinarily, ignorance of the law is a valid defense to criminal liability. (True/False)
-
The minimum acceptable standard for ethical behavior is compliance with the law.(True/False)
-
A release is an agreement in which one party gives up the right to pursue a legal claim against another party. (True/False)
-
In mediation, a mediator decides on the matter in dispute.(True/False)
-
Assertiveness & Self-Confidence Performance management Communication Strategies Agents 4 and 5 have been the ones with the lowest performance for 3rd week in a row. His Repeated Adjusted Positive...
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
Which of the following are chiral? (a) 2-Methyl-2,3-hexadiene (b) 4-Methyl-2,3-hexadiene (c) 2,4-Dimethyl-2,3-pentadiene
-
Show, by writing a suitable sequence of chemical equations, how you could prepare each of the following compounds from cyclopentene and any necessary organic or inorganic reagents: (a)...
-
Apply the knowledge gained by studying Table 20.2 to help you predict the major organic product obtained by reaction of benzoyl chloride with each of the following: (a) Benzoic acid (b) Ethanol (c)...
-
Approximately 50,000 new titles, including new editions, are published each year in the United States, giving rise to a $25 billion industry in 2001. In terms of percentage of sales, this industry...
-
When you think of political persuasion, you may think of the effortsthat political campaigns undertake to persuade you that their candidate is betterthan the other candidate. In truth, campaigns are...
-
Exeter, Inc., is a catalog firm that sells products in a number of different catalogs that it owns. The catalogs number in the dozens but fall into nine basic categories: 1. Clothing 2. Housewares 3....

Study smarter with the SolutionInn App



