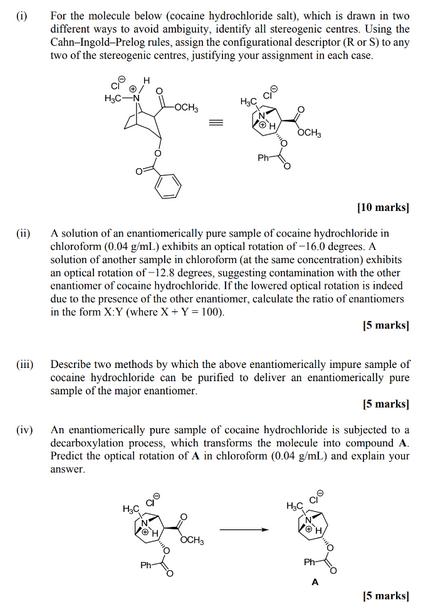
(i) For the molecule below (cocaine hydrochloride salt), which is drawn in two different ways to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(i) For the molecule below (cocaine hydrochloride salt), which is drawn in two different ways to avoid ambiguity, identify all stereogenic centres. Using the Cahn-Ingold-Prelog rules, assign the configurational descriptor (R or S) to any two of the stereogenic centres, justifying your assignment in each case. -OCH OCH (ii) [10 marks] A solution of an enantiomerically pure sample of cocaine hydrochloride in chloroform (0.04 g/ml.) exhibits an optical rotation of-16.0 degrees. A solution of another sample in chloroform (at the same concentration) exhibits an optical rotation of-12.8 degrees, suggesting contamination with the other enantiomer of cocaine hydrochloride. If the lowered optical rotation is indeed due to the presence of the other enantiomer, calculate the ratio of enantiomers in the form X:Y (where X + Y = 100). 15 marks] (iii) Describe two methods by which the above enantiomerically impure sample of cocaine hydrochloride can be purified to deliver an enantiomerically pure sample of the major enantiomer. (iv) [5 marks] An enantiomerically pure sample of cocaine hydrochloride is subjected to a decarboxylation process, which transforms the molecule into compound A. Predict the optical rotation of A in chloroform (0.04 g/mL) and explain your answer. OCH H,C 15 marks] (i) For the molecule below (cocaine hydrochloride salt), which is drawn in two different ways to avoid ambiguity, identify all stereogenic centres. Using the Cahn-Ingold-Prelog rules, assign the configurational descriptor (R or S) to any two of the stereogenic centres, justifying your assignment in each case. -OCH OCH (ii) [10 marks] A solution of an enantiomerically pure sample of cocaine hydrochloride in chloroform (0.04 g/ml.) exhibits an optical rotation of-16.0 degrees. A solution of another sample in chloroform (at the same concentration) exhibits an optical rotation of-12.8 degrees, suggesting contamination with the other enantiomer of cocaine hydrochloride. If the lowered optical rotation is indeed due to the presence of the other enantiomer, calculate the ratio of enantiomers in the form X:Y (where X + Y = 100). 15 marks] (iii) Describe two methods by which the above enantiomerically impure sample of cocaine hydrochloride can be purified to deliver an enantiomerically pure sample of the major enantiomer. (iv) [5 marks] An enantiomerically pure sample of cocaine hydrochloride is subjected to a decarboxylation process, which transforms the molecule into compound A. Predict the optical rotation of A in chloroform (0.04 g/mL) and explain your answer. OCH H,C 15 marks]
Expert Answer:

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemical engineering questions
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
Assignment: Based on your reading and analysis of the case study above, address the following items in a detailed essay response of approximately 600 words. Each number below should be addressed...
-
In answering the following questions, assume that the full deletion algorithm is used. Assume that merging is done when a bucket becomes empty. 1. Give an example of Extendible Hashing where deleting...
-
Sorrento Products uses a job costing accounting system for its manufacturing costs. A predetermined overhead rate based on machine-hours is used to apply overhead to individual jobs. An estimate of...
-
Tandrin Aviation Holdings Ltd. agreed to sell a jet aircraft to Aero Toy Store, LLC, for \($31.75\) million. ATS paid a \($3\) million deposit to a third party with the balance due upon delivery....
-
Premium Entries and Financial Statement Presentation Sycamore Candy Company offers a CD single as a premium for every five candy bar wrappers presented by customers together with $2.50. The candy...
-
Max Labs Inc. current portfolio of products has a 10% IRR and a 10% standard deviation. The correlation coefficients with the firm portfolio are 0.5 (r=0.5) for both, Dog Treats and Dog Food; zero...
-
The Metropolis Health Systems Rehabilitation and Wellness Center offers outpatient therapy and return-to-work services plus cardiac and pulmonary rehabilitation to get people back to a normal way of...
-
You may use outside sources in answering the following questions, . Remember to cite sources correctly if you do use them. using APA Format Open IT Savvy to pp. 152-154, The IT Savvy Monday Morning...
-
Patients have rights and expect a certain level of behavior from the people they are paying to treat them, in your role as a dental assistant list some of things that you are required to do for your...
-
Winey Cork Limited uses the allowance method for estimating uncollectible accounts. Required: Prepare journal entries to record the following transactions: January 5 Sold merchandise to Smith Inc for...
-
Read the Article: "Markets Kick Off 2023 in the Red" by Barron's. (link below)...
-
Spilly Ltd. uses the periodic inventory system and applied FIFO inventory costing. At the end of the annual accounting period, December 31, 2024, the accounting records for the best selling item in...
-
the estate of a grandmother provides 1/3 of the total for her oldest daughter, 1/5 of the total for her younger son, 1.6 of the total to her younger daughter, 1/10 of the total to charity, and the...
-
Does Outside Environmental Temperature Affect Oven Efficiency? Explain
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
The common practice of washing laboratory glassware with acetone can lead to unintended consequences. For example, a student plans to carry out the preparation of methylmagnesium iodide, CH3MgI,...
-
The science of pain control has evolved dramatically in the past several years. The body deals with pain by releasing anandamide (see Section 20-6). Anandamide binds to the cannabinoid receptor, the...
-
Rank each group of compounds in order of increasing boiling point, (a) Cyclohexane, cyclohexanol, chlorocyclohexane; (b) 2,3-dimethyl-2-pentanol, 2-methyl-2-hexanol, 2-heptanol.
-
Which of Chaterjis criticisms of MVO is/are valid? A. Only Criticism 1 B. Only Criticism 2 C. Both Criticism 1 and Criticism 2 Investment adviser Carl Monteo determines client asset allocations using...
-
Which of the characteristics put forth by Chaterji to describe the factor-based approach is/are correct? A. Only Characteristic 1 B. Only Characteristic 2 C. Both Characteristic 1 and Characteristic...
-
The change in the annual spending rate, in conjunction with the boards expectations regarding future enrollment and the need for endowment support, could justify that KUEs target weight for: A....

Study smarter with the SolutionInn App


