For the reaction NH4* (aq) + NO2 (aq)-> N2(g) + 2HO(g), if water is being produced...
Fantastic news! We've Found the answer you've been seeking!
Question:
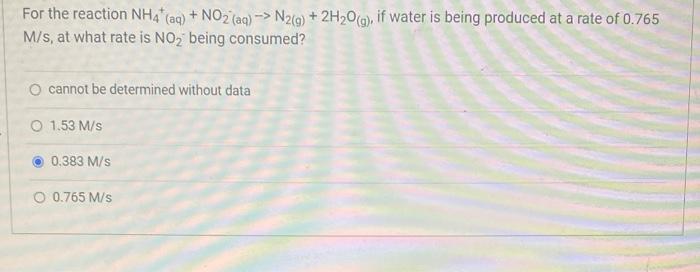
Transcribed Image Text:
For the reaction NH4* (aq) + NO2 (aq)-> N2(g) + 2H₂O(g), if water is being produced at a rate of 0.765 M/s, at what rate is NO₂ being consumed? O cannot be determined without data O 1.53 M/s 0.383 M/s O 0.765 M/s For the reaction NH4* (aq) + NO2 (aq)-> N2(g) + 2H₂O(g), if water is being produced at a rate of 0.765 M/s, at what rate is NO₂ being consumed? O cannot be determined without data O 1.53 M/s 0.383 M/s O 0.765 M/s
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below The balanced chemical equat... View the full answer

Posted Date:
Students also viewed these physics questions
-
Consider two identical spin particles in a harmonic oscillator potential. So that the Hamiltonian takes the form P? P?2 +mw?(r r2) 1 H = + 2m 2m Assume that the two-particle system has zero...
-
A harmonic oscillator potential of natural frequency v contains eight electrons and is in its lowest-energy state. (a) What is its energy? (b) What would the lowest energy be if the electrons were...
-
A harmonic oscillator potential with natural frequency contains N electrons and is in its state of lowest energy. Find expressions for the energy of the highest-energy electron for (a) N even (b) N...
-
Use the Chain Rule to calculate the partial derivatives. Express the answer in terms of the independent variables. OF -; F(u, v) = eu+v, u = x, v = xy
-
It was announced today that Florida, Inc., will acquire Menlo Park, Inc. Menlo Park has assets with a gross tax basis of $ 6 million and has $ 1.5 million of liabilities. Prior to being acquired,...
-
Fifty cities provided information on vacancy rates (in percent) for local apartments in the following frequency distribution. Vacancy Rate (in percent) ......Frequency 0 up to 3 .............. 5 3 up...
-
Home Realty, Incorporated, has been operating for three years and is owned by three investors. J. Doe owns 60 percent of the total outstanding stock of 9,000 shares and is the managing executive in...
-
1. Companies are developing ethical policies and guidelines for legal reasons, but also to clarify what is acceptable and what is not. Do you think any of the issues raised in the case required...
-
The output of the code below is #include #include int main(){ float x; x = floor(7.5); printf("%.2f ",x); return 0;}
-
Turn this orthographic drawing to isometric? 75 9.5 4.5 PLAN VIEW FRONT VIEVW RIGHT ELEVATION VIEW Figure I SS SZ SZT
-
Richard is a Native American Indian who has an enrollment number and is living on his tribe's reservation. Richard is a security guard for the tribal casino. In his spare time, Richard sells fishing...
-
Analyse the conceptual and operational difficulties surrounding the practice of strategic human resource planning;
-
Assume you have \(\$ 100,000\) to invest for one year, and decide to allocate \(60 \%\) to an all equity fund trading at \(\$ 400\) per share and \(40 \%\) to a bond fund trading at \(\$ 100\) per...
-
Review potential avenues for addressing the difficulties associated with human resource planning in order to enhance its operational viability.
-
An investor has a base \(10 \log\) utility function: \(U(x)=\log _{10}(x)\). Investment 1 has payoff \((1,100)\) with probabilities \((0.8,0.2)\). Investment 2 has payoff \((10,1000)\) with...
-
What factors have contributed to the relatively low level of importance attaching to management development?
-
Use the discriminant to determine the number of real solutions of the equation. 4x^(2)+3x+(13)/(2)=0 infinitely many real solutions two distinct real solutions one real solution no real solution 2...
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
A fi rm issues a bond with a coupon rate of 5.00% when the market interest rate is 5.50% on bonds of comparable risk and terms. One year later, the market interest rate increases to 6.00%. Based on...
-
A company issues 10,000,000 face value of 10-year bonds dated January 1, 2015 when the market interest rate on bonds of comparable risk and terms is 6%. Th e bonds pay 7% interest annually on...
-
Regarding a companys debt obligations, which of the following is most likely presented on the balance sheet? A . Eff ective interest rate. B . Maturity dates for debt obligations. C . Th e portion of...

Study smarter with the SolutionInn App

