Given: L-200 ft equal tangent parabolic curve with g = -1.25%, g = +1.25 %, VPI...
Fantastic news! We've Found the answer you've been seeking!
Question:
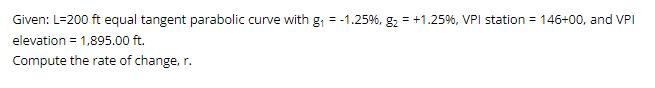
Transcribed Image Text:
Given: L-200 ft equal tangent parabolic curve with g₁ = -1.25%, g₂ = +1.25 %, VPI station = 146+00, and VPI elevation = 1,895.00 ft. Compute the rate of change, r. Given: L-200 ft equal tangent parabolic curve with g₁ = -1.25%, g₂ = +1.25 %, VPI station = 146+00, and VPI elevation = 1,895.00 ft. Compute the rate of change, r.
Expert Answer:
Answer rating: 100% (QA)
Answer Explanation i Loss on sale of investment Cost value of investment sold Sale val... View the full answer

Posted Date:
Students also viewed these accounting questions
-
Write a code that includes CSS and html to create a web page, provided that the page has two interfaces.
-
9. Suppose we are solving for Arb with A E Rmxn being rank deficient (minimum norm solution) (a) For b(A) why wouldn't the solution (ATA)-Ab work? (b) Suppose beR(A). Show that the error vector is...
-
For blood circulation in a specific human body artery, the following data is given, Average inner diameter of the artery = 6.8 mm Average pressure gradient per meter of length = 75 pa 1- Find the...
-
Suppose the European Parliament is considering legislation that will decrease tariffs on agricultural products imported from the United States. The benefit of the tariff reduction is estimated at 5...
-
On May 1, 2016, Taft Company acquires a 80% interest in Marcus Company for $400,000. The fair value of the NCI is $100,000. The following determination and distribution of excess schedule is...
-
The information in Figure 8-20 is for the employees of the Marcia Felix Corporation. a. Use a DBMS software package to create a database for this figure. b. What is the average pay rate for these...
-
How can a paralegal assist a lawyer with the voir dire of the jury?
-
Starbucks is hoping to make use of its excess restaurant capacity in the evenings by experimenting with selling beer and wine. It speculates that the only additional costs are hiring more of the same...
-
FINANCIAL RISK MANAGRMENT Protecting Interest Income/Revenue From the banker's point of view, when the banker quotes a floating interest, in doing so, the banker is passing on the interest rate risk...
-
I See The Light Projected Income Statement For the Period Ending December 31, 20x1 $ 1,125,000.00 25,000 lamps @ $45.00 @ $30.00 Sales Cost of Goods Sold 750.000.00 Gross Profit $ 375.000.00 Selling...
-
Convert the (010) and (101) planes into the four-index miller-bravais scheme for hexagonal unit cells. Draw the same planes within a hexagonal unit cell.
-
Sharrer Inc.s only temporary difference at the beginning and end of 2015 is caused by a \($2\) million deferred gain for tax purposes for an installment sale of a plant asset, and the related...
-
Refer to the note issued by Coldwell, Inc. in BE14-9. During 2015, Coldwell experiences financial difficulties. On January 1, 2016, Coldwell negotiates a settlement of the note by issuing to Flint...
-
At December 31, 2015, Conway Corporation had a defined benefit obligation of 510,000 and plan assets of 322,000. Prepare a pension reconciliation schedule for Conway.
-
Suppose the option in the previous question is a put option rather than a call option. a. What is the intrinsic value for this put option? b. What is the time premium for this put option? Data from...
-
The following defined pension data of Yang Corp. apply to the year 2015 (amounts in thousands). Instructions For 2015, prepare a pension worksheet for Yang Corp. that shows the journal entry for...
-
Colin is 40 years old and wants to retire in 27 years. His family has a history of living well into their 90s. Therefore, he estimates that he will live to age 95. He currently has a salary of...
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
The following experimental data are available for \(\mathrm{CO}_{2}\) gas at \(92^{\circ} \mathrm{C}\) : PR 1 2 3 4 6 8 10 0.856 0.583 0.535 0.620 0.800 0.975 1.160 Find the fugacity of CO2 at 100...
-
What do you mean by extent of reaction? Derive an expression of the relationship between mole fraction of the component and the extent of reaction.
-
Give an informatory note on equilibrium constant of the chemical reaction.

Study smarter with the SolutionInn App

