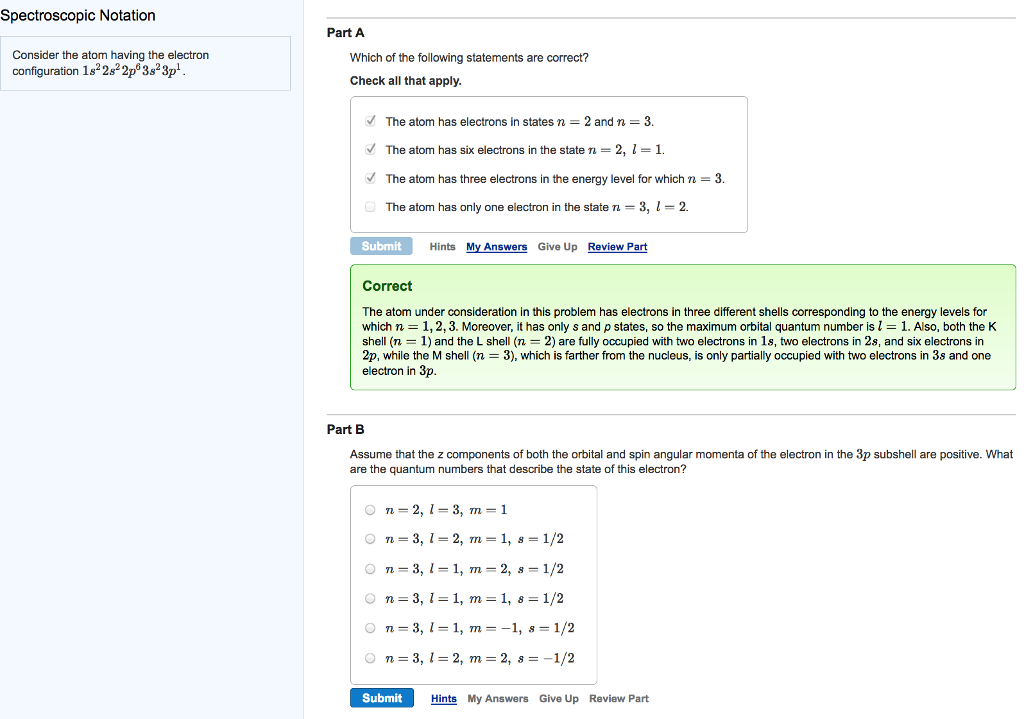
Spectroscopic Notation Consider the atom having the electron configuration 182s2p 3s3p. Part A Which of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Spectroscopic Notation Consider the atom having the electron configuration 18²2s²2p 3s²3p¹. Part A Which of the following statements are correct? Check all that apply. ✔The atom has electrons in states n = 2 and n = 3. ✔The atom has six electrons in the state n = 2, l = 1. ✔The atom has three electrons in the energy level for which n = 3. The atom has only one electron in the state n = 3, l = 2. Submit Hints My Answers Give Up Review Part Correct The atom under consideration in this problem has electrons in three different shells corresponding to the energy levels for which n = 1, 2, 3. Moreover, it has only s and p states, so the maximum orbital quantum number is 1 = 1. Also, both the K shell (n = 1) and the L shell (n = 2) are fully occupied with two electrons in 1s, two electrons in 2s, and six electrons in 2p, while the M shell (n = 3), which is farther from the nucleus, is only partially occupied with two electrons in 3s and one electron in 3p. Part B Assume that the z components of both the orbital and spin angular momenta of the electron in the 3p subshell are positive. What are the quantum numbers that describe the state of this electron? On=2, 1-3, m = 1 On 3, 1-2, m = 1, s = 1/2 On=3, 11, m2, s = 1/2 On=3, 1=1, m = 1, 8 = 1/2 On 3, 1-1, m = -1, s = 1/2 n = 3, 12, m = 2, s = -1/2 Submit Hints My Answers Give Up Review Part Spectroscopic Notation Consider the atom having the electron configuration 18²2s²2p 3s²3p¹. Part A Which of the following statements are correct? Check all that apply. ✔The atom has electrons in states n = 2 and n = 3. ✔The atom has six electrons in the state n = 2, l = 1. ✔The atom has three electrons in the energy level for which n = 3. The atom has only one electron in the state n = 3, l = 2. Submit Hints My Answers Give Up Review Part Correct The atom under consideration in this problem has electrons in three different shells corresponding to the energy levels for which n = 1, 2, 3. Moreover, it has only s and p states, so the maximum orbital quantum number is 1 = 1. Also, both the K shell (n = 1) and the L shell (n = 2) are fully occupied with two electrons in 1s, two electrons in 2s, and six electrons in 2p, while the M shell (n = 3), which is farther from the nucleus, is only partially occupied with two electrons in 3s and one electron in 3p. Part B Assume that the z components of both the orbital and spin angular momenta of the electron in the 3p subshell are positive. What are the quantum numbers that describe the state of this electron? On=2, 1-3, m = 1 On 3, 1-2, m = 1, s = 1/2 On=3, 11, m2, s = 1/2 On=3, 1=1, m = 1, 8 = 1/2 On 3, 1-1, m = -1, s = 1/2 n = 3, 12, m = 2, s = -1/2 Submit Hints My Answers Give Up Review Part
Expert Answer:
Related Book For 

Basic Marketing Research
ISBN: 978-1133188544
8th edition
Authors: Tom J. Brown, Tracy A. Suter, Gilbert A. Churchill
Posted Date:
Students also viewed these chemistry questions
-
muy al nord Jong orit bas 19. Suppose that there are n electrons (n <2N) in the allowed band described in the preceding problem. Show that the Fermi energy of the electron distrubution at absolute...
-
I need the answer as soon as possible. Halsey Corporation first issued stock on January 1, 2016. Halsey has the following stock outstanding on December 31, 2019: Preferred Stock (5 percent...
-
9. Suppose we are solving for Arb with A E Rmxn being rank deficient (minimum norm solution) (a) For b(A) why wouldn't the solution (ATA)-Ab work? (b) Suppose beR(A). Show that the error vector is...
-
Bob was waiting at the crosswalk for the light to turn green. As he stood there, a car that was stopped in the road next to him suddenly exploded, and Bob was injured by the blast. A defect in the...
-
The following are comparative financial statements of the Perez Company for 2006, 2007, and 2008: Required On the basis of the given information: 1. Prepare horizontal analyses for Perez Company...
-
A relatively new term that has been introduced to the Web environment is Web service. What is a Web service and how does it differ from the concept of a Web application?
-
List the things that should not be done in a fraud investigation.
-
Accountants for Carlson, Inc. have assembled the following data for the year ended December 31, 2016: Prepare Carlsons statement of cash flows using the indirect method. Include an accompanying...
-
answer the ff. Question 31 31. The estimated ending inventory is Question 32 32. The amount of inventory shortage or overage is . (please put a negative sign if it is an inventory shortage). D Ques...
-
Putt Corporation acquired 70 percent of Slice Companys voting common stock on January 1, 20X3, for $158,900. Slice reported common stock outstanding of $100,000 and retained earnings of $85,000. The...
-
Watch my sisters keeper Your approved movie must focus on death, dying or grief and be amajor component of the movie. As you watch this movie consider how the death topics are being portrayed and...
-
In the liabilities section of its 2008 balance sheet, Wells Fargo & Company reported "noninterestbearing deposits" of over \(\$ 150\) billion. Wells Fargo is a very large banking company. In the...
-
If you were a compensation consultant to a company, how would you recommend it provide incentives to the CE O and other executives? Why?
-
Whitewater Corporation borrowed \(\$ 40,000\) from the bank on October 1, 2011. The note had a 9 percent annual rate of interest and matured on March 31, 2012. Interest and principal were paid in...
-
The College Book Mart sells books and other supplies to students in a state where the sales tax rate is 8 percent. The College Book Mart engaged in the following transactions for 2011. Sales tax of 8...
-
What is meant by goods flow and cost flow?
-
The Question is: ABC Research projects a return on equity of 30%. Management plans to plow back 30% of all earnings into the firm. Earnings this year will be $5 per share, and investors expect a 15%...
-
Explain the circumstances that could result in a long-term bank loan being shown in a statement of financial position as a current liability.
-
The marketing manager at Crystallo Bottling Company is studying the relationship between the number of cases per shipment and transportation costs. An analysis of the data from the previous problem...
-
Compute the 95 percent confidence interval for the percentage of respondents who had traveled more than 250 miles to visit the theme park using the information in the previous question.
-
For each of the following situations, identify the appropriate target population and sampling frame. a. A local chapter of the American Lung Association wants to test the effectiveness of a brochure...
-
Answer the same question as in the previous problem but for the scheme n+1 u+ - u i At un i+1 1 -u-1 +c. = 0. 2Ax
-
Modify the scheme (7.39) for the Burgers equation by changing the central difference approximation of the convective term to the upwind approximation. Assume that \(u\) is always positive. How does...
-
Repeat Problem 7.4 for the scheme based on the Runge-Kutta method of the second order (7.51).

Study smarter with the SolutionInn App



