Consider the following reaction: HO (1) H(g) + O (g) AH = +285.8 kJ mol-...
Fantastic news! We've Found the answer you've been seeking!
Question:
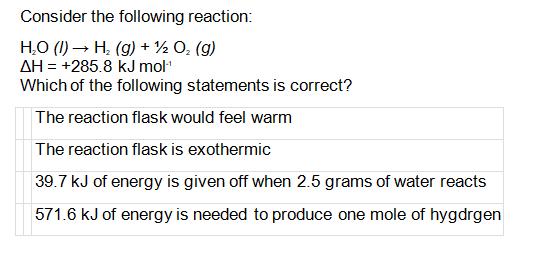
Transcribed Image Text:
Consider the following reaction: H₂O (1)→ H₂(g) + ½ O₂ (g) AH = +285.8 kJ mol-¹ Which of the following statements is correct? The reaction flask would feel warm The reaction flask is exothermic 39.7 kJ of energy is given off when 2.5 grams of water reacts 571.6 kJ of energy is needed to produce one mole of hygdrgen Consider the following reaction: H₂O (1)→ H₂(g) + ½ O₂ (g) AH = +285.8 kJ mol-¹ Which of the following statements is correct? The reaction flask would feel warm The reaction flask is exothermic 39.7 kJ of energy is given off when 2.5 grams of water reacts 571.6 kJ of energy is needed to produce one mole of hygdrgen
Expert Answer:
Answer rating: 100% (QA)
The reaction is endothermic because the enthalpy change is positive ... View the full answer

Related Book For 

Ethics in Accounting A Decision Making Approach
ISBN: 978-1118928332
1st edition
Authors: Gordon Klein
Posted Date:
Students also viewed these chemistry questions
-
What do you think about the idea of codes of conduct? Give three reasons why an organization ought to have a code of conduct, and give three reasons why an organization should not have a code of...
-
What do you think about Wholesomes idea for opening a new distribution channel?
-
You have a very outgoing personality and are extremely friendly with a staff member named Adelana. Adelana reports directly to you in the company's Billing Department. You are a CPA, but Adelana is...
-
A particle of mass m moves along the x-axis under a force F = ??m?2x. The amplitude is b. Show that the period is given by 4. T= dx, (11.83) where (11.84) Y =1+ 2c2 (2-2).
-
What is an exemption? How many exemptions may a taxpayer claim?
-
Use the Dahamel integral method to derive expressions for the response of an undamped system subjected to the forcing functions shown in Figs. 4.46(a). F(t) F(t) F(t) Fo(1 COS 210 Fo Fo Fo to 0 to 0...
-
Construct an environmental profile for a company of your choice. How might the profile be of use to management?
-
The administrator of elections for the city of Crossville has been asked to perform an activity analysis of its optical scanning center. The optical scanning center reads voter forms into the...
-
Missing Data ABC Itd selected data for the month of August related to current year are presented below (in millions) Beginning Work in process Inventory Beginning Direct Material Inventory Direct...
-
You work in the human resources department of your company helping new employees fill out the necessary paperwork to get their first paycheck. There are a number of decisions that employees must make...
-
The assets and liabilities of a company are $128,000 and $84,000, respectively. Owner's equity should equal Oa. $84,000 Ob. $128,000 O c. $44,000 O d. $212,000
-
The following was submitted under the original assignment of "write small program that uses dynamic binding".. now what is requested is to suggest another way to take advantage of the dynamic biding...
-
Productivity can also be described as the effectiveness with which the resources input in a plant are translated to customer satisfaction-oriented products. Explain
-
Alex worked for years for MegaCorp. During his employment with MegaCorp, he learned a great deal of confidential information and knew that if this information got into the hands of competitors or the...
-
Maxey & Sons manufactures two types of storage cabinets-Type A and Type Band applies manufacturing overhead to all units at the rate of $152 per machine hour. Production information follows....
-
Suppose S 0 SF / $ = SF 0 . 9 4 9 4 / $ and F 6 - monthSF / $ = SF 0 . 9 4 7 8 / $ . What is the percentage forward premium / discount on the U . S . dollar?
-
Piedmont Company segments its business into two regions-North and South. The company prepared the contribution format segmented income statement as shown: Sales Variable expenses Contribution margin...
-
31. What is the income that can be received over 15 years from $500,000 earning 6% annually? 32. What is the semiannual payment required to retire $50,000 in debt over 5 years at 8% compounded...
-
As a student, you have the opportunity to work in the Volunteer Income Tax Assistance Program, commonly known as VITA. VITA volunteers principally prepare tax returns for indigent taxpayers, free of...
-
Section 5061(a) of California's Business and Professions Code states that "a person engaged in the practice of public accountancy shall not ... pay a fee or commission to obtain a client." You are a...
-
Before issuance, most corporate and governmental bonds receive a credit rating that usually ranges from a high of triple-A down to B-. Nearly all bond ratings are issued by one of three credit rating...
-
The following data are taken from the comparative balance sheets of Girard Billiards Club, which prepares its financial statements using the accrual basis of accounting. Fees are billed to members...
-
Colin Mochrie Company has the following balances in selected accounts on December 31, 2008. All the accounts have normal balances. Colin Mochrie Company debits prepayments to expense accounts when...
-
Tony Masasi started his own consulting firm, Masasi Company, on June 1, 2008. The trial balance at June 30 is shown. In addition to those accounts listed on the trial balance, the chart of accounts...

Study smarter with the SolutionInn App


